| Author | Affiliation |
|---|---|
| Getaw Worku Hassen, MD, PhD | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
| Albert Hwang, BS | Columbia University Post-baccalaureate Pre-medical Program, School of General Studies, New York, New York |
| Lydia Liyun Liu, BA, MA | Columbia University Post-baccalaureate Pre-medical Program, School of General Studies, New York, New York |
| Felicia Mualim, BS | Columbia University Post-baccalaureate Pre-medical Program, School of General Studies, New York, New York |
| Toshiro Sembo, | Columbia University Post-baccalaureate Pre-medical Program, School of General Studies, New York, New York |
| Ting Jia Tu, BS, MS | Rowan University School of Osteopathic Medicine, Stratford, New Jersey |
| Daniel Hsiang Wei, BS, MS | Columbia University Post-baccalaureate Pre-medical Program, School of General Studies, New York, New York |
| Paul Johnston, MPH | Columbia University Post-baccalaureate Pre-medical Program, School of General Studies, New York, New York |
| Ana Costea, MD | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
| Carlos Meletiche, MD | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
| Shakeel Usmani, MD | Lutheran Medical Center, Department of Emergency Medicine, Brooklyn, New York |
| Andre Barber, | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
| Rajnish Jaiswal, MD | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
| Hossein Kalantari, MD, MPH | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
Introduction
Methods
Results
Discussion
Limitations
Conclusion
ABSTRACT
Introduction: Contrast-induced nephropathy (CIN), defined as an increase in serum creatinine (SCr) greater than 25% or ≥0.5 mg/dL within 3 days of intravenous (IV) contrast administration in the absence of an alternative cause, is the third most common cause of new acute renal failure in hospitalized patients. It is known to increase in-hospital mortality up to 27%. The purpose of this study was to investigate the rate of outpatient follow up and the occurrence of CIN in patients who presented to the emergency department (ED) and were discharged home after computed tomography (CT) of the abdomen and pelvis (AP) with IV contrast.
Methods: We conducted a single center retrospective review of charts for patients who required CT of AP with IV contrast and who were discharged home. Patients’ clinical data included the presence of diabetes mellitus, hypertension, chronic kidney disease (CKD) and congestive heart failure (CHF).
Results: Five hundred and thirty six patients underwent CT of AP with IV contrast in 2011 and were discharged home. Diabetes mellitus was documented in 96 patients (18%). Hypertension was present in 141 patients (26.3%), and 82 patients (15.3%) were on angiotensin-converting-enzyme inhibitors (ACEI). Five patients (0.9%) had documented CHF and all of them were taking furosemide. Seventy patients (13%) had a baseline SCr >1.2 mg/dL. One hundred fifty patients (28%) followed up in one of the clinics or the ED within one week after discharge, but only 40 patients (7.5%) had laboratory workup. Out of 40 patients who followed up within 1 week after discharge, 9 patients (22.5%) developed CIN. One hundred ninety patients (35.4%) followed up in one of the clinics or the ED after 7 days and within 1 month after discharge, but only 71 patients (13.2%) had laboratory workup completed. Out of 71 patients who followed up within 1 month, 11 patients (15%) developed CIN. The overall incidence of CIN was 15.3% (17 out of 111 patients).
Conclusion: There was a poor outpatient follow up after CT of AP with IV contrast and biochemically CIN appears to be present in some patients. Unlike previous reports that CKD is the major risk factor for CIN, our results demonstrated that risk factors such as advanced age, DM and hypertension seem to predispose patients to CIN rather than abnormal baseline SCr. [West J Emerg Med. 2014;15(3):276–281.]
INTRODUCTION
Contrast-induced nephropathy (CIN), defined as an increase in serum creatinine (SCr) greater than 25% or ≥0.5 mg/dL within 3 days of IV contrast administration in the absence of an alternative cause, is the third most common cause of new acute renal failure in hospitalized patients.1–3 Usually CIN is diagnosed by serial laboratory examination in hospitalized patients.4–7 The SCr level returns within 1 to 3 weeks to baseline or a new baseline on serial follow up, and CIN is believed to resolve within 3 weeks.8 The overall incidence of CIN is estimated to be 4.96% even if it varied based on the presence of various risk factors.9,10 In general, CIN is known to increase in-hospital mortality up to 27%.1,5 Hospitalized patients are subjected to serial laboratory examination, and once they develop CIN specialists such as nephrologists evaluate and advise on the management. In addition, nephrotoxic drugs are withheld and the patients’ fluid status is monitored and adjusted. To monitor for development of CIN some authorities recommend measuring the SCr repeatedly for more than 48 hours after administration of intravenous (IV) contrast.11 Patients that are discharged from the ED following the administration of IV contrast for computed tomography (CT) of abdomen and pelvis (AP) are not subjected to serial laboratory examination, including SCr. Hence, the incidence and outcomes of CIN in these patients are unknown. Moreover, the fluid intake and medication compliance in these patients are not regulated or monitored after discharge.
The incidence of CIN in an outpatient setting has been studied prospectively by Mitchell et al.12 Their study ensured regular follow up with a team that followed patients for the purpose of the study. Our study focused on a population with low socio-economic status, no regular primary care physician, and poor clinic follow up.
We investigated the rate of outpatient follow up and incidence of CIN in patients who had been discharged from the ED after undergoing CT of AP with administration of IV contrast. The purpose of this retrospective study was to investigate the rate of outpatient follow up and the incidence of CIN in patients who presented to the ED, received CT of AP with IV contrast and were discharged home. Particularly noted were patients with underlying congestive heart failure (CHF), hypertension and diabetes mellitus (DM). These conditions were considered as risk factors and were used as data collection elements with a plan to test to see if they contributed to the development of CIN in our population.
METHODS
We conducted a retrospective review of charts from patients who presented to our ED with conditions requiring CT of AP with IV contrast and who were discharged home from January 1, 2011, to December 31, 2011. This review was approved by the institutional review board. We conducted the study in a single urban academic center with annual visit approximately 70,000 patients. Patients were identified using current procedural terminology (CPT) codes for CT of AP with IV contrast. All patients who received CT in the ED and were subsequently discharged were selected for the study. We reviewed the electronic patient charts for demographics, number of CTs, laboratory results, disposition, clinic follow up, medication use and co-morbidities. Two independent groups reviewed the charts and adjusted missing or conflicting data as needed. Patients who had conflicting or missing data were removed from the study. Records were reviewed to see whether these patients followed up in one of the specialty clinics, the medical clinic or the ED within 1 week and after 1 week, but within 1 month from discharge after undergoing CT of AP with IV contrast in the ED. We noted baseline kidney function on the day of their ED visit and results on subsequent visits. We defined CIN as an increase in Scr greater than 25% or ≥0.5 mg/dL from the base line as it was accepted in the literature.9
Our institution uses two types of IV contrast medium: [Omnipaque (Iohexol) for patients with SCr less than 1.5 mg/dl and Visipaque (Iodixanol) for those with SCr 1.5–2.0 mg/dl)]. The bolus dose of the contrast medium is either 120 ml or 150 ml of the corresponding contrast agent, depending on the weight of the patient. Patients who weigh >210 pounds (lbs) or approximately 95 kilograms (Kg) receive 150 ml and patients who weigh 180 lbs (81 Kg)-210 lbs (95 Kg) receive 120 ml. Patients who weigh less than 180 lbs (81Kg) receive 1.5 ml/Kg.
Finally, we analyzed the results using descriptive statistics Software SPSS 13. Chi square analysis was used to evaluate the proportion of patients that developed CIN after the use of IV contrast.
RESULTS
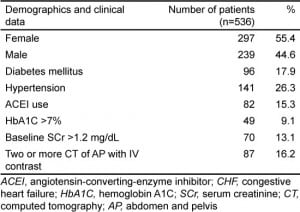
Five hundred thirty-six patients underwent CT of AP with IV contrast in 2011 and were discharged home. Two hundred ninety-seven patients (55.4%) were females and 239 (44.6%) were males. In the prospective study by Mitchell et al the mean age for developed CIN was 54 years.12 We arbitrarily chose the age of 50 as the threshold to stratify age as a risk factor for developing CIN. Three hundred and eighteen of the patients (59.3%) were younger than 50 years old and 218 (40.7%) were older than 50. Diabetes mellitus was documented in 96 patients (18%). Hypertension was present in 141 patients (26.3%) and 82 patients (15.3%) were on angiotensin-converting-enzyme inhibitors (ACEI). Five patients (0.9%) had documented CHF and all of them were taking furosemide. Four hundred sixty-six patients (87%) had a baseline Scr <1.2 mg/dL and 70 (13%) had a baseline SCr >1.2 mg/dL. Sixty-one patients (11%) had an estimated glomerular filtration rate (eGFR) <60 mL/min/1.73 m2 of which 49 (80.3%) had a Scr <1.5 mg/dL. Eighty-seven patients (16.2%) underwent 2 or more CT AP with IV contrast within 1 year. Forty nine patients (9.1%) had a hemoglobin A1C (HbA1C) level >7% as a marker of poorly controlled DM.
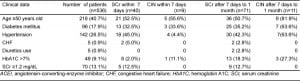
One hundred fifty patients (28%) followed up in one of the clinics or the ED within 1 week after discharge, but only 40 patients (7.5%) had laboratory workup. Out of 40 patients who followed up within one week after discharge, 9 patients (22.5%) developed CIN. One hundred ninety patients (35.4%) followed up in one of the clinics or the ED after 1 week and within 1 month after discharge, but only 71 patients (13.2%) had laboratory workup completed. Out of 71 patients who followed up after 1 week and within 1 month, 11 patients (15%) developed CIN. Of all patients who had laboratory tests on follow up visits, 17/111 patients (15.3%) patients were found to have an elevated SCr. Of the 9 CIN patients who followed up within 1 week after discharge 4 patients (44.4%) came back to the clinic within 1 month. Three of them (75%) continued to have CIN biochemically. One of them (25%) had worsening of the renal function (from 18% on 1 week follow up to 55% on 1 month follow up) and the renal function of the second patient stayed reduced at 25%. Two other patients (50%) improved their renal function. One of them improved from 88% to 13% and the other one from 200% to 150%, but remained to have CIN biochemically.
Demographics and clinical data of patients are listed in Tables 1 and 2.
DISCUSSION
The incidence of CIN has been studied in hospital patients with underlying medical conditions.4,13–16 Admitted patients are usually subjected to serial laboratory examination, fluid input and output monitoring, and care is taken to not expose them to medications that could cause kidney injury. The same might not be true for discharged patients, as their fluid intake is not regulated and they may potentially resume nephrotoxic medication, such as certain antibiotics or non-steroidal anti-inflammatory drugs (NSAIDs), either by not following instructions or for lack of proper instruction.
The exact mechanism leading to CIN is not clear, but combinations of toxic and ischemic injury to tubular cells are suggested as contributory factors. The proposed mechanism for contrast-induced nephropathy include increased fluid viscosity secondary to the contrast agent concentration due to medullar hyperosmolar environment, which leads to decreased flow in the medullary tubules and vessels.17–21 The reduced flow leads to increased contact time of contrast medium and tubular cells and subsequent production of radical oxygen species resulting in cytotoxic damage.22,23 Direct cytotoxic effect of contrast medium on tubular cells is also one of the mechanisms suspected to cause tubular cell injury.19 In addition, medullary vasoconstriction causes hypoxic cellular injury.20,24,25 The presence of risk factors is likely to contribute to and/or augment the kidney injury.18,26 The effects and incidence of CIN were studied prospectively in the ED setting in patients who underwent CT of the chest with IV contrast.12,27–29 These studies revealed a rate of CIN up to 12%. Hospitalized patients who undergo CT of AP may receive pre-procedural hydration for up to 12 hours when needed. In emergency situations there is not much time to hydrate patients for 12 hours prior to obtaining CT. This puts discharged ED patients at greater risk for developing CIN after receiving IV contrast for CT of AP, particularly in patients with underlying medical conditions such as DM and hypertension. The majority of patients in our area have no primary care physician and they rarely use the medical clinic for follow up.
The best strategy for management of CIN is to avoid its occurrence. Therefore, it is of paramount importance to identify patients at risk using a simple questionnaire regarding underlying medical conditions and nephrotoxic drug usage. Scoring systems have been developed to predict the risk for developing CIN30,31 These scoring systems may be used to identify patients at risk for developing CIN in the ED. The likelihood of developing CIN can be estimated by the number of risk factors present before the administration of IV contrast. Patient-related risk factors are divided into major (preexisting renal disease and DM) and minor (advanced age, female gender, hypertension and nephrotoxic drugs).11 Although the incidence of CIN is low in patients with normal renal function, the incidence may be as high as 25% in patients with preexisting renal impairment or other risk factors, such as DM, CHF, advanced age, and concurrent use of nephrotoxic drugs.2
The risk assessment for developing CIN can be made qualitatively based on the risk factors and quantitatively with blood urea nitrogen (BUN) and SCr. The risk of CIN increases with the number of risk factors. Preexisting renal impairment is an independent risk factor and risk predictor for CIN.32–34 In addition to preexisting renal impairment other risk factors for developing CIN are thought to be advanced age, CHF, DM and dehydration.32,35–37
It has been shown in 1 model that the risk of developing CIN was relatively constant at baseline SCr level <1.1 mg/dL, but increased sharply at levels >1.2 mg/dL.13 According to the CIN Consensus Working Panel the risk of CIN is elevated and becomes clinically important when the baseline SCr level is ≥1.3 mg/dL in men and ≥1.0 in women, equivalent to estimated eGFR <60 mL/min per 1.75 m2.8,33 Unfortunately SCr lacks the sensitivity to identify clinically significant CIN and some recommend using eGFR as a better marker to identify CIN.6 A recent study demonstrated that the commonly used SCr cutoff of 1.5 mg/dL for IV contrast administration fails to identify up to 40% of the ED patients at risk for CIN.38 In addition, important measures to minimize CIN are volume expansion before the procedure, adequate fluid intake after the procedure, avoidance of nephrotoxic drug use and early follow up to assess renal function in high risk patients.
Outpatient clinic follow up is an essential part of patient management and continuity of care. Noncompliance with follow up in the outpatient clinic is a well-known problem worldwide. Both patient and hospital-related factors, such as transport constraints and crowding, play a role in delayed or absence of outpatient follow up.39–43 Close follow up is very important in high risk patients in order to identify complications at early stages and treat them appropriately. Failure to follow up can have serious consequences particularly in patients with underlying medical conditions, such as preexisting renal insufficiency, CHF, DM and hypertension.39
Our retrospective study showed that a significant portion of patients in our community did not follow up for evaluation and blood testing after discharge from the ED. The overall incidence of CIN after CT of AP with IV contrast was 15.3% (17 patients out of 111). We had 2 groups (follow up within a week and follow up after a week until 1 month after discharge). The second group was to see if the CIN had resolved or progressed. The true incidence is unknown due to poor follow up, inclusion of patients with only CT of AP with IV contrast and missing data from patients who followed up outside of our hospital. It is not obvious whether the biochemical changes as reflected in worsening SCr have any clinical significance. A mortality from CIN was reported in the Mitchell et al study.27,28 In our study relevant clinical information, such as vomiting, nausea, altered mental status or uremia, was not recorded at the clinic visit. Eighty-seven patients (16.2%) had 2 or more CT of AP with IV contrast within 1 year. Of these 87 patients, 5 (5.7%) developed CIN within 1 week and 6 additional patients (6.9%) developed CIN within a 1-month period. Thirty three percent of the patients who developed CIN in both follow ups were older than 50, had DM and received 2 or more CT of AP with IV contrast within 1 year. The likelihood of developing CIN increases with the number of risk factors.
This retrospective study highlights that a fair number of patients with risk factors for developing CIN were discharged from the ED after receiving CT of AP with IV contrast. Only a few followed up in the clinic, and some developed CIN (at least biochemically, Tables 1 and 2). Without long term follow up with SCr, or better with eGFR measurements and clinical evaluation of discharged patents, it is difficult to estimate the true incidence and the outcomes of CIN. A prospective study with long term follow up with SCr and clinical evaluation of high risk patients may guide the future approach regarding CIN in patients undergoing CT of AP with IV contrast in the ED.
The likelihood of developing CIN increases with the number of risk factors.32,33,44,45 Patients can be stratified for risk of developing CIN based on the number of risk factors they have at the first ED visit. Further management recommendations should be made based on preexisting risk factors, the laboratory results and clinical evaluation at the first follow up visit.
A prospective study assessing both clinical parameters and biochemical changes may give a better picture of CIN in patients discharged from ED after CT of AP with IV contrast. In addition, information with regard to the amount of fluid intake and whether or not they have stopped taking nephrotoxic drugs should be obtained.
One possible intervention would be to identify high risk patients based on a scoring system in the ED and develop a callback system to ensure these patients return to either the ED or to one of our hospitals’ clinics for follow up and laboratory testing to assess kidney function on days 2 or 3. Patients should receive clear instruction about the need to follow up after receiving CT of AP with IV contrast.
LIMITATIONS
The study is limited by small sample size due to lack of follow up. It is a single center retrospective study with lack of some relevant information due to lack of documentation. In addition, our study focused on CT of AP and did not include patients who had CT of other parts of the body, such as the chest and head, and we did not track whether patients had other nephrotoxins or CT of other organ systems during the study period that may have accounted for CIN without our knowledge. We followed up patients just for 1 month. The results of the short-term follow up might not reflect the natural course of CIN. Furthermore, the laboratory results of patients who followed up in their primary care physicians’ office or other hospitals were not included in our study. It was not clear from the chart review whether or not hypotensive episodes were present in patients with CIN as well as the amount of fluid intake after discharge. The role of other potential causes of nephropathy, such as CHF and nephrotoxic drugs especially long term, could not be studied in this setting. Moreover, patients received 2 different types of contrast based on their baseline SCr and different doses based on their bodyweight. The effect of contrast medium amount and contrast medium type used on pathogenesis of CIN is largely unknown in this cohort.
CONCLUSION
Biochemical CIN appears to be present after CT of AP with IV contrast in some patients who followed up. It was not obvious whether the biochemical changes caused clinically significant symptoms in these patients. Unlike previous reports that chronic kidney disease is the major risk factor for CIN, our results demonstrated that risk factors such as advanced age, DM and hypertension seem to predispose patients to CIN rather than abnormal baseline SCr. In order to make meaningful conclusions a multi-center prospective study with larger sample size is necessary.
Footnotes
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
Supervising Section Editor: Sanjay Arora, MD
Address for Correspondence: Getaw Worku Hassen, MD, PhD. New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, 1901 First Ave, New York, NY 10028. Email: getawh@yahoo.com. 5 / 2014; 15:276 – 281
Submission history: Revision received April 9, 2013; Submitted July 21, 2013; Accepted August 4, 2013
Full text available through open access at http://escholarship.org/uc/uciem_westjem
REFERENCES
1. Nash K, Hafeez A, Hou S. Hospital-acquired renal insufficiency. Am J Kidney Dis. 2002; 39:930-936
2. McCullough PA, Wolyn R, Rocher LL Acute renal failure after coronary intervention: incidence, risk factors, and relationship to mortality. Am J Med. 1997; 103:368-375
3. Rudnick MR, Goldfarb S, Wexler L Nephrotoxicity of ionic and nonionic contrast media in 1196 patients: a randomized trial. The Iohexol Cooperative Study. Kidney Int. 1995; 47:254-261
4. Best PJM, Lennon R, Ting HH The impact of renal insufficiency on clinical outcomes in patients undergoing percutaneous coronary interventions. J Am Coll Cardiol. 2002; 39:1113-1119
5. Levy EM, Viscoli CM, Horwitz RI. The effect of acute renal failure on mortality. A cohort analysis. JAMA. 1996; 275:1489-1494
6. McCullough P. Outcomes of contrast-induced nephropathy: experience in patients undergoing cardiovascular intervention. Catheter Cardiovasc Interv. 2006; 67:335-343
7. McCullough PA, Adam A, Becker CR Epidemiology and prognostic implications of contrast-induced nephropathy. Am J Cardiol. 2006; 98:5K-13K
8. Berns AS. Nephrotoxicity of contrast media. Kidney Int. 1989; 36:730-740
9. Kooiman J, Pasha SM, Zondag W Meta-analysis: serum creatinine changes following contrast enhanced CT imaging. Eur J Radiol. 2012; 81:2554-2561
10. Moos SI, van Vemde DN, Stoker J Contrast induced nephropathy in patients undergoing intravenous (IV) contrast enhanced computed tomography (CECT) and the relationship with risk factors: A meta-analysis. Eur J Radiol. 2013; 82:e387-399
11. Gami AS, Garovic VD. Contrast nephropathy after coronary angiography. Mayo Clin Proc. 2004; 79:211-219
12. Mitchell AM, Jones AE, Tumlin JA Incidence of contrast-induced nephropathy after contrast-enhanced computed tomography in the outpatient setting. Clin J Am Soc Nephrol. 2010; 5:4-9
13. Davidson CJ, Hlatky M, Morris KG Cardiovascular and renal toxicity of a nonionic radiographic contrast agent after cardiac catheterization. A prospective trial. Ann Intern Med. 1989; 110:119-124
14. From AM, Bartholmai BJ, Williams AW Mortality associated with nephropathy after radiographic contrast exposure. Mayo Clin Proc. 2008; 83:1095-1100
15. Ghani AA, Tohamy KY. Risk score for contrast induced nephropathy following percutaneous coronary intervention. Saudi J Kidney Dis Transpl. 2009; 20:240-245
16. Gleeson TG, Bulugahapitiya S. Contrast-induced nephropathy. Am J Roentgenol. 2004; 183:1673-1689
17. Persson PB, Hansell P, Liss P. Pathophysiology of contrast medium-induced nephropathy. Kidney Int. 2005; 68:14-22
18. Seeliger E, Sendeski M, Rihal CS Contrast-induced kidney injury: mechanisms, risk factors, and prevention. Eur Heart J. 2012; 33:2007-2015
19. Sendeski MM. Pathophysiology of renal tissue damage by iodinated contrast media. Clin Exp Pharmacol Physiol. 2011; 38:292-299
20. Tumlin J, Stacul F, Adam A Pathophysiology of contrast-induced nephropathy. Am J Cardiol. 2006; 98:14K-20K
21. Jorgensen AL. Contrast-induced nephropathy: pathophysiology and preventive strategies. Crit Care Nurse. 2013; 33:37-46
22. Liss P, Nygren A, Olsson U Effects of contrast media and mannitol on renal medullary blood flow and red cell aggregation in the rat kidney. Kidney Int. 1996; 49:1268-1275
23. Nygren A. Contrast media and regional renal blood flow. A study of the effects of ionic and non-ionic monomeric and dimeric contrast media in the rat. Acta Radiol Suppl. 1992; 378:123-135
24. Solomon R, Deray G. How to prevent contrast-induced nephropathy and manage risk patients: practical recommendations. Kidney Int. 2006; :S51-53
25. Rauch D, Drescher P, Pereira FJ Comparison of iodinated contrast media-induced renal vasoconstriction in human, rabbit, dog, and pig arteries. Invest Radiol. 1997; 32:315-319
26. Pucelikova T, Dangas G, Mehran R. Contrast-induced nephropathy. Catheter Cardiovasc Interv. 2008; 71:62-72
27. Mitchell AM, Jones AE, Tumlin JA Prospective study of the incidence of contrast-induced nephropathy among patients evaluated for pulmonary embolism by contrast-enhanced computed tomography. Acad Emerg Med. 2012; 19:618-625
28. Mitchell AM, Jones AE, Tumlin JA Immediate complications of intravenous contrast for computed tomography imaging in the outpatient setting are rare. Acad Emerg Med. 2011; 18:1005-1009
29. Mitchell AM, Kline JA. Contrast nephropathy following computed tomography angiography of the chest for pulmonary embolism in the emergency department. J Thromb Haemost. 2007; 5:50-54
30. Rabbani AB, Nallamothu BK. Contrast-induced nephropathy risk assessment in real world practice. Am J Med. 2011; 124:1127-1128
31. Mehran R, Aymong ED, Nikolsky E A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: development and initial validation. J Am Coll Cardiol. 2004; 44:1393-1399
32. McCullough PA, Adam A, Becker CR Risk prediction of contrast-induced nephropathy. Am J Cardiol. 2006; 98:27K-36K
33. Lameire N, Adam A, Becker CR Baseline renal function screening. Am J Cardiol. 2006; 98:21K-26K
34. Lameire NH. Contrast-induced nephropathy–prevention and risk reduction. Nephrol Dial Transplant. 2006; 21:i11-23
35. Kamdar A, Weidmann P, Makoff DL Acute renal failure following intravenous use of radiographic contrast dyes in patients with diabetes mellitus. Diabetes. 1977; 26:643-649
36. Nakamura Y, Yoshida T, Yamamoto M Exacerbation of renal dysfunction following contrast media study in diabetic patients. Jpn J Med. 1981; 20:188-193
37. Dudzinski PJ, Petrone AF, Persoff M Acute renal failure following high dose excretory urography in dehydrated patients. J Urol. 1971; 106:619-621
38. Band RA, Gaieski DF, Mills AM Discordance between serum creatinine and creatinine clearance for identification of ED patients with abdominal pain at risk for contrast-induced nephropathy. Am J Emerg Med. 2007; 25:268-272
39. Griffin SJ. Lost to follow-up: the problem of defaulters from diabetes clinics. Diabet Med. 1998; 15:S14-24
40. Renders CM, Valk GD, Griffin S Interventions to improve the management of diabetes mellitus in primary care, outpatient and community settings. Cochrane Database Syst Rev. 2001; :CD001481
41. Ross JD, McIver A, Blakely A Why do patients default from follow-up at a genitourinary clinic: a multivariate analysis. Genitourin Med. 1995; 71:393-395
42. Van der Meer G, Loock JW. Why patients miss follow-up appointments: a prospective control-matched study. East Afr J Public Health. 2008; 5:154-156
43. Cheung PT, Wiler JL, Lowe RA National Study of Barriers to Timely Primary Care and Emergency Department Utilization Among Medicaid Beneficiaries. Ann Emerg Med. 2012; 60:4-10
44. Chong E, Poh KK, Liang S Risk factors and clinical outcomes for contrast-induced nephropathy after percutaneous coronary intervention in patients with normal serum creatinine. Ann Acad Med Singapore. 2010; 39:374-380
45. Rich MW, Crecelius CA. Incidence, risk factors, and clinical course of acute renal insufficiency after cardiac catheterization in patients 70 years of age or older. A prospective study. Arch Intern Med. 1990; 150:1237-1242


