| Author | Affiliations |
| Daniel J. Shogilev, MD | Duke University, Division of Emergency Medicine, Duke University, Durham, North Carolina |
| Nicolaj Duus, MD | Beth Israel Deaconess Medical Center, Department of Emergency Medicine, Boston, Massachusetts |
| Stephen R. Odom, MD | Beth Israel Deaconess Medical Center, Department of Surgery, Boston, Massachusetts |
| Nathan I. Shapiro, MD, MPH | Beth Israel Deaconess Medical Center, Department of Emergency Medicine and Center for Vascular Biology, Boston, Massachusetts |
Introduction
Methods
Conclusion
ABSTRACT
Introduction
Acute appendicitis is the most common abdominal emergency requiring emergency surgery. However, the diagnosis is often challenging and the decision to operate, observe or further work-up a patient is often unclear. The utility of clinical scoring systems (namely the Alvarado score), laboratory markers, and the development of novel markers in the diagnosis of appendicitis remains controversial. This article presents an update on the diagnostic approach to appendicitis through an evidence-based review.
Methods
We performed a broad Medline search of radiological imaging, the Alvarado score, common laboratory markers, and novel markers in patients with suspected appendicitis.
Results
Computed tomography (CT) is the most accurate mode of imaging for suspected cases of appendicitis, but the associated increase in radiation exposure is problematic. The Alvarado score is a clinical scoring system that is used to predict the likelihood of appendicitis based on signs, symptoms and laboratory data. It can help risk stratify patients with suspected appendicitis and potentially decrease the use of CT imaging in patients with certain Alvarado scores. White blood cell (WBC), C-reactive protein (CRP), granulocyte count and proportion of polymorphonuclear (PMN) cells are frequently elevated in patients with appendicitis, but are insufficient on their own as a diagnostic modality. When multiple markers are used in combination their diagnostic utility is greatly increased. Several novel markers have been proposed to aid in the diagnosis of appendicitis; however, while promising, most are only in the preliminary stages of being studied.
Conclusion
While CT is the most accurate mode of imaging in suspected appendicitis, the accompanying radiation is a concern. Ultrasound may help in the diagnosis while decreasing the need for CT in certain circumstances. The Alvarado Score has good diagnostic utility at specific cutoff points. Laboratory markers have very limited diagnostic utility on their own but show promise when used in combination. Further studies are warranted for laboratory markers in combination and to validate potential novel markers.
INTRODUCTION
Acute appendicitis is the most common abdominal emergency requiring surgery with an estimated lifetime prevalence of 7%.1 Despite its high prevalence, the diagnosis of appendicitis remains challenging. The diagnosis of appendicitis embodies Sir William Osler’s spirit when he stated, “Medicine is a science of uncertainty and an art of probability.” The clinical presentation is often atypical and the diagnosis is especially difficult because symptoms often overlap with other conditions.2 The fundamental clinical decision in the diagnosis of a patient with suspected appendicitis is whether to operate or not. Ideally, the goal is to expeditiously treat all cases of appendicitis without unnecessary surgical interventions. A 2001 study reported negative appendectomy rates between 15% and 34% with approximately 15% being commonly accepted as appropriate to reduce the incidence of perforation.3,4
The meaningful evaluation of acute appendicitis balances early operative intervention in hopes of preventing perforation against a more restricted approach with the hope of reducing the risk of unnecessary surgery. Additionally, physicians must consider the accuracy, delay-to-surgery, and radiation risks of using computed tomography (CT) imaging, as well as the reliability of laboratory results and clinical scoring systems. Lastly, physicians’ actions are often unfortunately influenced by malpractice litigation as appendicitis is one of the most frequent medical conditions associated with litigation against emergency department (ED) physicians with claims paid to patients in up to one third of cases.5,6
The goal of this article is to present the reader with an update on the diagnostic approach to appendicitis by providing an evidence-based review of radiological imaging, clinical scoring systems, laboratory testing, and novel biomarkers for appendicitis.
METHODS
We did a broad PubMed search using the follow key phrases: “diagnosis of appendicitis”, “imaging AND appendicitis”, “CT AND appendicitis”, “US AND appendicitis” , “laboratory markers in appendicitis”, “Alvarado score” and “novel markers in appendicitis.” We searched meta-analysis, systematic reviews, reviews and clinical trials dating back to 2000. Only published research was used in our paper. We also conducted a secondary source search on the most relevant articles. Since many meta-analyses are available, we focus on these, but also include relevant single publication data. Our focus is on bringing the reader up to date in this rapidly evolving field.
Radiological Imaging
Technological advances and an increase in availability of CT have fundamentally changed the approach to appendicitis. In a 2011 study of 2,871 patients, multi-detector CT had a sensitivity of 98.5% and a specificity of 98%.7 Similarly, another 2006 meta-analysis consisting of data from 31 studies and 4341 patients yielded both a sensitivity and specificity of 94%.8 A 2011 meta-analysis made up of 28 studies comprising 9,330 patients found that the negative appendectomy rate was 8.7% when using CT compared to 16.7% when using clinical evaluation alone.9 Similarly, this study also demonstrated a significantly higher negative appendectomy rate during the pre-CT era compared to the post-CT era (10% vs. 21.5%).9 Interestingly, the incidence of appendiceal perforation, the most significant complication of appendicitis, was reported as unchanged by the use of CT.9
A 2007 systematic review (25 studies and 9,121 patients) examining ultrasound (US) in the diagnosis of equivocal acute appendicitis yielded a sensitivity of 83.7% and a specificity of 95.9%.10 Similarly, a 2006 meta-analysis found similar results in both children and adults.8 A long-standing criticism of US use in the diagnosis of appendicitis is that it is less accurate than CT and user-dependent. This criticism was evident in a pooled study of surgeon-performed US imaging in cases of suspected appendicitis where considerable inter- and intra-observer heterogeneity was seen. While this was largely attributed to the operator-dependent variability of US, it also likely reflected the resolution spectrum seen in bedside ultrasound machines compared to higher-resolution machines in radiology departments.11
The improvement in diagnostic accuracy with widespread adoption of CT for appendicitis comes with the concerns over increased radiation exposure and long-term cancer risks. One study estimated that there would be approximately 29,000 future cancers related to CTs performed in the year 2007, alone, with the largest proportion coming from scans of the abdomen/pelvis, the scan of choice in suspected appendicitis.12 Another study estimated that there would be the development of a radiation-associated cancer for every 620 males and every 470 females who received abdomen/pelvic CT with contrast at the age of 20. For those undergoing this type of CT at the age of 60, it was estimated that 1 in 1,250 males and 1 in 1,320 females would develop CT radiation-associated cancers.13
Efforts to avoid the disadvantages of CT while maintaining diagnostic accuracy are warranted. One popular strategy, particularly in the pediatric population, is to perform US as the initial radiologic step in the diagnosis of appendicitis because of ability to “rule in” appendicitis if positive. If clear signs of appendicitis are present, then surgery is performed without the need for CT. Due to US’s limited sensitivity, a negative or equivocal result cannot be used to “rule out” appendicitis, and CT is employed. Poortman et al. followed this protocol in their study of 151 patients with suspected appendicitis. US was positive for 79 patients, and 71 of them had acute appendicitis. Those who had a negative or inconclusive US, underwent CT, of which 21 were positive for appendicitis (verified in surgery).14 In a large pediatric study of 620 children with equivocal US findings, some patients received a follow-up CT while others were observed. Here, there were no known missed diagnosis of appendicitis.15 More recently, a 2014 study of 662 patients under 18 with suspected appendicitis compared CT with a radiation free US-magnetic resonance imaging (MRI) protocol (similar to that of US-CT protocol). They found this US-MRI protocol and CT to have no significant differences in time to antibiotic administration, time to appendectomy, negative appendectomy rate, perforation rate or length of stay.16 In-depth research to help discern an optimal strategy combining US, CT and even MRI is warranted since it has the potential of reducing costs and radiation exposure while maintaining a low negative appendectomy rate.
Efforts have also been made to limit CT’s high radiation levels with low-dose CT imaging. Kim et al. examined the use of this low-dose abdominal CT for evaluating suspected appendicitis. In their single-center study of 891 adolescents and young adults, they demonstrated that low dose CT (which used 1/4th of the standard radiation) and standard CT had a similar negative appendectomy rates and no major differences in perforation rates.17 Other smaller studies have yielded similar results,18,19 and there is currently at least one large, multicenter randomized control trial underway.20
The Alvarado Scoring System
The Alvarado score is a clinical scoring system used to stratify the risk of appendicitis in patients presenting with abdominal pain. Alvarado’s original work was published in 1988 and is based on his retrospective data analysis of 305 patients presenting with abdominal pain suggestive of acute appendicitis. This study found eight predictive factors of diagnostic value in acute appendicitis and assigned each factor a value of 1 or 2 based on their diagnostic weight. A score of 1 was given for each of the following: elevated temperature >37.3°C, rebound tenderness, migration of pain to right lower quadrant (RLQ), anorexia, nausea or vomiting, and leukocyte left shift. A score of 2 was given for RLQ tenderness and leukocytosis >10 000. The likelihood of appendicitis and specific management recommendations are given based on the total score. A score of 5 or 6 is “compatible” with the diagnosis of acute appendicitis and recommends the clinician observe or serially examine the patient. A score of 7 or 8 is “probable” appendicitis and a score of 9 or 10 is “very probable” appendicitis and recommends surgical intervention.21
Ironically, the results in subsequent validation studies of the Alvarado score largely outperform the original study’s findings and provide the major support for consideration of the rule in clinical practice. In a meta-analysis by Ohle et al.22 conducted in 2011, a review of 29 studies including 5,960 subjects revealed that for a cutoff of 5 (criteria to observe/admit) there was a sensitivity of 99% (95% CI: 97-99%) and specificity of 43% (36-51%). At a cutoff of 7 (criteria to proceed directly to surgery) sensitivity was 82% (76-86%) and specificity was 81% (76-85%). Based on these results, the authors argue that using a cutoff score of 5 or lower provides a good “ruling out” score, while a cutoff of 7 is not sufficiently specific enough to provide an adequate “ruling in” score.22 However, several other smaller studies did not find such a high sensitivity. A 2007 retrospective study of 150 patients aged 7 and older who presented to the ED with abdominal pain found that 5% of patients with a score of 3 or less had appendicitis, as did 36% of patients with a score between 4-6.23 Similarly, in a retrospective study of 215 adults and children who presented with acute abdominal pain, Gwynn et al.1 found that 8.4% (12 of 143) of subjects with appendicitis had an Alvarado score below 5. Another retrospective study of 156 children found that 9% of subjects with complicated appendicitis would have been overlooked with the use of the Alvarado score.24
What can we conclude from this? Based on the results of the aforementioned 2011 meta-analysis we can conclude that since an Alvarado score of ≥5 had a sensitivity of 99%, this is a promising strategy for ruling out patients who are clinically at low risk for appendicitis. These patients may be observed clinically or with outpatient warnings as opposed to receiving an ED CT. Conversely, the specificity of the Alvarado score does not reliably determine the need for surgery without further clinical assessment and testing. While some smaller studies call into question the accuracy of the Alvarado score, this approach does seem to be a reasonable starting point in the assessment of a patient with suspected appendicitis.
One main critique of the Alvarado score is its applicability in pediatric populations. A meta-analysis revealed that at a cutoff of 5 (observe/admit criteria) for 1,635 pediatric patients there was a sensitivity of 99% and specificity of 57%, similar to that of the adult subjects. At a cutoff of 7 (surgery criteria) sensitivity was 87% and specificity was 76%.22 Thus, similar clinical prediction rules can be prescribed as above. This meta-analysis did not give a clear definition of what constitutes a “child,” and thus, it is not clear whether these results apply to all pediatric populations. The Alvarado score requires children to identify migration of pain, nausea and anorexia, variables that are not easily identified by very young children.25
Laboratory Markers
White Blood Cells (WBCs):
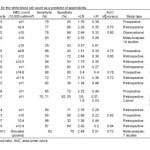
The degree of white blood cell (WBC) elevation in acute appendicitis has been extensively studied. It is very commonly elevated in patients with acute appendicitis. However, it is not a specific marker and is commonly elevated in patients with other inflammatory conditions included in the differential diagnosis.26 Table 1 lists 19 publications including two meta-analyses of WBC sensitivity, specificity, likelihood ratios and accuracy. Part of the difficulty in drawing precise conclusions from these data is that there is great variability in the WBC concentration cut-offs. A WBC cut-off of greater than 10-12 000 cell/mm3 yielded a range of sensitivity between 65-85% and specificity between 32-82%.2,25,27-36 A 2003 meta-analysis including 14 studies (3,382 patients) likely gives a representative approximation of the true sensitivity and specificity of a WBC>10 000 cell/mm3 measured at 83% and 67%, with a positive and negative likelihood ratio of 2.52 and 0.26.2 Thus, on its own, a WBC>10 000 cell/mm3 is insufficient as a diagnostic modality. The discriminatory power of an elevated WBC count, expressed as area under the curve (AUC), is shown in Table 1. The AUC values range from 0.72 to 0.8 reflecting WBC’s modest discriminatory power.28,31,34,36-42 The likelihood ratio values are also shown in Table 1. While these values are statistically significant, it is generally agreed that clinically significant values for likelihood ratios are either greater than 10 or under 0.1. Thus, these values only alter the probability of the diagnosis to a modest degree, and thus, do not change the diagnostic workup on its own.
C-Reactive Protein (CRP):
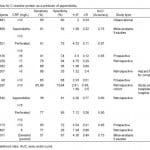
CRP is an acute phase reactant. Its diagnostic significance is largely based on both its kinetic properties and its utility as a marker for complicated/advanced appendicitis. CRP levels show an increase between 8-12 hours after the onset of inflammatory processes with a peak between 24 and 48 hours, which is later than that of WBC.43,44 Consequently, CRP contributes little diagnostic utility early in the case of simple appendicitis. Table 2 lists 12 studies including two large meta-analyses on CRP levels in appendicitis. A CRP cut-off of >10 mg/L yielded a range of sensitivity between 65-85% and a specificity between 59-73%.2,28,33,34,36,45 In a study of 542 people the AUC of CRP on day 1 was only 0.60 compared to 0.77 on day 2 and 0.88 day 3. In cases of perforated appendicitis, the AUC was 0.90 on day 1, 0.92 on day 2 and 0.96 on day 3.44 Thus, CRP serves as a strong predictor for appendiceal perforation but is quite limited for appendicitis in general.44,46
Granulocyte Count and Proportion of Polymorphonuclear (PMN) Cells:
Table 3 lists 10 publications including one meta-analysis of granulocyte count and proportion of polymorphonuclear (PMN) that document their sensitivity, specificity, likelihood ratios and accuracy. A normal granulocyte count ranges between 2500-6000 cells. A modestly elevated PMN greater than 7-7.5 x109 cells/L yielded a range of sensitivity of 71-89% and a specificity of 48-80% in diagnosis of acute appendicitis.2,28,37,39,45 Andersson’s 2003 meta-analysis of several laboratory variables in acute appendicitis showed that a granulocyte count greater than 11×109/L had a larger likelihood ratio than any other lab marker measured and was one of the strongest laboratory discriminators of appendicitis.2 Yet, the likelihood ratios were not even increased to near clinically significant levels unless the PMN was very elevated to >13 x 109 cells/L. At this level, 2 studies of 502 and 1013 patients found a likelihood ratio of 7.09 and 6.67, respectively.2,39
Similarly, as detailed in Table 3, PMN ratio>75%, was also a discriminator of acute appendicitis but had limited clinical significance due to a sensitivity of 66–87% and specificity of 33-84%.2,28,32,34,39,41,47 Again, the likelihood ratios were not high enough to significantly change the probability of appendicitis.
Assessing “left shift,” defined as a band form count >700/microL, a 2002 retrospective study of 1013 subjects found a sensitivity of 28%, a specificity of 87%, and an AUC of 0.58.39 In this study the presence of “left shift” provided a likelihood ratio of 2.17 which has limited clinical significance. In another study of 722 patients with a mean age of 9.7 years who presented to the pediatric ED, a “left shift” had a total sensitivity of 59% and a specificity 90%.48 Here the likelihood ratio was 5.7. Thus, while possibly providing some diagnostic clues, “left shift” did not yield any absolute conclusions.
Temperature:
History of fever provides very little diagnostic significance in acute appendicitis.2,28,38,49 In a study of 492 patients, a temperature of greater than 37.7°C had a sensitivity of 70% and a specificity of 65%.28 In a meta-analysis consisting of 570 patients with suspected appendicitis, history of fever only gave a likelihood ratio of 1.64.2 A meta-analysis of 502 patients reported that the average measured temperature in non-surgical abdominal pain was 37.7°C versus 37.8°C in cases of appendicitis on first measurement.28 However, the diagnostic significance of the variable increased significantly on serial examination and was an important discriminator of advanced appendicitis. The receiver operating characteristic (ROC) curve for all appendicitis on primary examination was 0.56 increasing to 0.77 after serial assessment.28 Thus, although initial temperature does not provide much diagnostic value initially, it still remains a parameter worth looking at when observing someone with suspected appendicitis.
Lab Values in Combination:
Perhaps a multi-marker approach is necessary for the diagnosis of appendicitis. While the individual elements of clinical and laboratory data carry limited diagnostic value, many studies show an exponential increase in the predictive and discriminatory power when multiple markers are combined.35,41,50-54 The major limitation of these publications is that some of the data may have been over-fit and the product of post-hoc analyses.
Table 4 shows many of the different combinations of lab markers evaluated. A study of 502 patients over the age of 10 years found that the combined inflammatory parameters (including WBC, PMN cells, PMN ratio, body temperature and CRP) had an accuracy of 0.85, which was similar to the total accuracy of clinical findings (0.87) and greater than all elements of disease history (0.78).28 The largest study identified included 897 patients and gave a sensitivity of 99% and specificity of 6% when any one of CRP≥8 mg/L, WBC≥10.4 x 103 cells/mm3 or PMN Ratio >74% was present, and a sensitivity of 98% and specificity of 12% when either WBC or CRP was elevated.47 Thus, while the absence of all of these laboratory markers can potentially “rule out” the diagnosis of appendicitis, the complete lack of specificity severely limits its application. In a smaller study of 98 patients, Sengupta et al.33 calculated a sensitivity of 100%, a specificity of 50% and a NPV of 100% when either CRP≥10 mg/L or WBC≥11 x 103 cells/mm3. Vaughn-Shaw et al.45 replicated Sengupta’s study in 297 patients and found a lower sensitivity of 92-94% and a specificity of 60-64%. In another prospective study of 102 patients (49 who had appendicitis), the combined AUC of a WBC>109 cells/L and CRP>6 mg/L was 0.96 with a likelihood ratio of 23.32 when all variables were present, 0.53 when at least one variable was present and 0.03 when all variables were absent.2 Many of these studies used different cut-off points so it is difficult to compare them. However, based on these results, acute appendicitis is very unlikely when WBC, CRP and PMN ratio are all within normal limits. As such, if a patient presents with clinical findings of appendicitis but no elevation of any of these lab markers, based on a collection of very small studies, the diagnosis of appendicitis is unlikely. While these studies are further limited by secondary and post-hoc analyses, they do provide some empiric evidence for a multi-marker approach. However, further validation is warranted.
Table 4 also shows the sensitivities and specificities when both CRP and WBC or CRP, WBC and PMN ratio are all elevated. Again, different combinations and cut-off values were used making it difficult to compare individual studies. The data show a specificity that ranges from 77-99%.33-35,45,55 Thus, because of this wide range of measured values, the current studies do not show a consistently high enough specificity to “rule in” appendicitis and warrant surgery without further clinical and/or imaging workup.
Interestingly, in a 1999 study by Gronroos et al.,52 none of the 200 consecutive adult patients with acute appendicitis had both CRP and WBC within normal range. However, Gronroos53 also reported that in the same study done on children, normal values of both WBC and CRP were found in 7 of 100 consecutive children. Therefore, the sensitivity and specificity of combined laboratory markers may vary in different age demographics.
This analysis is limited by the relative scarcity and small sample sizes of the literature examining the combined use of markers. Small studies have shown enhanced diagnostic potential and utility but much larger sample sizes are needed before any absolute recommendations can be made. Furthermore, evaluation of this multi-marker approach in different patient demographic groups, most notably children, adults and the elderly is warranted.
Novel Markers – Diagnostic Markers of the Future?
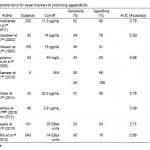
In response to the difficulty of making the accurate diagnosis of appendicitis and to decrease CT utilization and negative appendectomy rates, there has been much effort to search for novel markers. Table 5 lists several of these markers, which we will now briefly discuss. While some of these markers have shown early promise, the power of these studies is limited due to the small sample size.
Interleukin 6 (IL-6) is a cytokine that plays a focal role in the activation of the acute inflammatory response.40,56,57 A 2011 prospective study of 280 patients aged 3-18 with suspected appendicitis showed the IL-6 levels increase early appendicitis, and mean concentration also increases with the degree of inflammation.40,56,57 The sensitivity and specificity at different cut-off points are shown in Table 5. In a small prospective study of 80 patients, Paajanen et al.43 found the sensitivity, specificity and accuracy of IL-6 to be higher than that of WBC or CRP. While these studies show a clear relationship between IL-6 levels and acute appendicitis, they did not show that IL-6 improved the diagnosis of appendcitis.40,43,58,59
Serum Amyloid A (SAA) is a non-specific inflammatory marker. A small 2005 study of 42 patients with a mean age of 10.6 years and confirmed appendicitis on surgery calculated a sensitivity of 86%, a specificity of 83% and an AUC of 0.96 at a cutoff of SAA>45 mg/L. All 42 patients with acute appendicitis had elevated SAA levels, whereas only 14/42 had normal WBC values and 9/42 had normal CRP values.60 They also found that SAA had an early and more dynamic increase in inflammatory conditions compared to that of WBC and CRP. Thus, SAA may be useful in early appendicitis.
Muenzer et al..61 studied leukocyte gene expression (Riboleukograms) and cytokine profiles in children being evaluated for appendicitis. In a training cohort of 20 patients, they first identified 28 genes and five cytokines that were strongly associated with the diagnosis of appendicitis. They subsequently tested the diagnostic potential of these genes and cytokines in eight patients. Four out of the five patients with confirmed appendicitis would have been correctly diagnosed using riboleukograms alone. Out of the three patients without appendicitis, there would have been one false-positive result. Using the five identified plasma cytokines alone, only one out of four patients with appendicitis was correctly identified. However, all three patients without appendicitis were correctly identified here. Thus, riboleukograms showed potential for being a sensitive marker and plasma cytokines as a specific marker for acute appendicitis. Some of the major limitations of this study are the very small sample size, cost and real-time technical feasibility.
Allister et al.62 tested the utility of Granulocyte colony-stimulating factor (G-CSF) in the diagnosis of acute appendicitis in 32 patients with a mean age of 12 years. G-CSF is over-expressed in acute appendicitis and acts on the bone marrow to stimulate the production and release of granulocytes into the peripheral blood. Using a cut-off of 28.3 pg/ml yielded a sensitivity of 91% and a specificity of 51%. Additionally, serum levels of G-CSF closely correlated with the severity of inflammation and thus have the potential to complement other diagnostic measures while also helping to determine the severity of acute appendicitis.
Another promising novel marker in acute appendicitis is urine Leucine-rich α-2-glycoprotein (LRG). LRG is believed to be shed earlier in the urine of patients than locally activated neutrophils. A 2010 and 2012 study by Kentsis et al.63,64 of 49 patients found LRG detected more than 100-fold in the urine of patients under 18 years with appendicitis compared to those without. It was found to be elevated in the urine of some patients with acute appendicitis even in the absence of macroscopic changes. (Two patients had a pathologic appendix but had negative imaging were identified by elevated LRG levels.) It is also likely increased in pyelonephritis and other bacterial infections. Direct measurement of urine LRG using a select ion monitoring mass spectrometry assay yielded an AUC of 0.99, but using a commercially available LRG-ELISA the AUC was only 0.80 due to an immunoassay interference effect. One of the major focuses moving forward is to see if elevated urine LRG is sufficiently sensitive and specific enough to effect the clinical decision making of ED physicians. Furthermore, more effort is required in order to develop a standard widespread available laboratory technique that is able to accurately measure LRG.
Calprotectin (also known as S100A8/A9) is a calcium-binding protein associated with acute inflammation, specifically of gastrointestinal origin. The relationship between Calprotectin as a diagnostic tool in acute appendicitis was first studied by Bealer et al.65 in 2010. In a preliminary study of 181 patients, Bealer found a sensitivity of 93% and a specificity of 54% at a cut-off of 20 Elisa units. Their promising results persuaded the investigators to undergo a larger ongoing study examining this relationship. In 2012, Mills et al.66 conducted a similar study with 843 patients. Using a cut-off of 14 Elisa units yielded a sensitivity of 96% and a specificity of 16%. One major problem in this study was that the measured value of ELISA for Calprotectin showed a 13-43% increase in actual levels due to a shipping effect whereby the test values were inflated due to delay in analysis. Thus, the next step in development of Calprotectin as a lab marker for acute appendicitis is to create an accurate real-time laboratory analyzer. In short, Calprotectin is a promising new marker of appendicitis that may help differentiate acute appendicitis from non-inflammatory causes of acute abdominal pain.
CONCLUSION
The purpose of this article was to present the reader with an update on the diagnostic approach to suspected appendicitis by providing an evidence-based review of radiological imaging, clinical scoring systems, laboratory testing, and novel biomarkers for appendicitis. CT remains the best radiological modality for diagnosing appendicitis but radiation exposure and long term cancer risks are a major concern. The alternative use of ultrasound may help decrease the use of CT in certain circumstances. In adults, an Alvarado score up to five shows promise at ruling out appendicitis. Laboratory markers all contribute to the presentation of appendicitis but are unable to change the diagnostic management of suspected appendicitis on their own. When used in combination they show greater promise. Lastly, there are several novel markers that have showed early promise in diagnostic capability of suspected appendicitis. Further exploration of some of these markers, as well as potential others, is warranted.
ACKNOWLEDGEMENTS
Footnotes
Supervising Section Editor: Eric R. Snoey, MD
Full text available through open access at http://escholarship.org/uc/uciem_westjem
Address for Correspondence: Daniel Shogilev, MD, 4130 Garrett Rd. Apt. 1014, Durham, North Carolina, 27707. E-mail: dshogilev@gmail.com.
Submission history: Submitted February 19, 2014; Revision received July 29, 2014; Accepted September 2, 2014
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
- Gwynn LK. The diagnosis of acute appendicitis: clinical assessment versus computed tomography evaluation. J Emerg Med. 2001;21(2):119-123.
- Andersson RE. Meta-analysis of the clinical and laboratory diagnosis of appendicitis. Br J Surg. 2004;91(1):28-37.
- Bergeron E. Clinical judgment remains of great value in the diagnosis of acute appendicitis. Can J Surg. 2006;49(2):96-100.
- Flum DR, Morris A, Koepsell T, et al. Has misdiagnosis of appendicitis decreased over time? A population-based analysis. JAMA. 2001;286(14):1748-1753.
- Brown TW, McCarthy ML, Kelen GD, et al. An epidemiologic study of closed emergency department malpractice claims in a national database of physician malpractice insurers. Acad Emerg Med. 2010;17(5):553-560.
- Howell JM, Eddy OL, Lukens TW, et al. Clinical policy: critical issues in the evaluation and management of emergency department patients with suspected appendicitis. Ann Emerg Med. 2010;55(1):71-116.
- Pickhardt PJ, Lawrence EM, Pooler BD, et al. Diagnostic performance of multidetector computed tomography for suspected acute appendicitis. Ann Intern Med. 2011;154(12):789-796,W-291.
- Doria AS, Moineddin R, Kellenberger CJ, et al. US or CT for Diagnosis of Appendicitis in Children and Adults? A Meta-Analysis. Radiology. 2006;241(1):83-94.
- Krajewski S, Brown J, Phang PT, et al. Impact of computed tomography of the abdomen on clinical outcomes in patients with acute right lower quadrant pain: a meta-analysis. Can J Surg. 2011;54(1):43-53.
- Al-Khayal KA, Al-Omran MA. Computed tomography and ultrasonography in the diagnosis of equivocal acute appendicitis. A meta-analysis. Saudi Med J. 2007;28(2):173-180.
- Carroll PJ, Gibson D, El-Faedy O, et al. Surgeon-performed ultrasound at the bedside for the detection of appendicitis and gallstones: systematic review and meta-analysis. Am J Surg. 2013;205(1):102-108.
- Berrington de Gonzalez A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
- Smith-Bindman R, Lipson J, Marcus R, et al. Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med. 2009;169(22):2078-2086.
- Poortman P, Oostvogel HJ, Bosma E, et al. Improving diagnosis of acute appendicitis: results of a diagnostic pathway with standard use of ultrasonography followed by selective use of CT. J Am Coll Surg. 2009;208(3):434-441.
- Ramarajan N, Krishnamoorthi R, Gharahbaghian L, et al. Clinical correlation needed: what do emergency physicians do after an equivocal ultrasound for pediatric acute appendicitis? J Clin Ultrasound. 2014;42(7):385-94.
- Aspelund G, Fingeret A, Gross E, et al. Ultrasonography/MRI versus CT for diagnosing appendicitis. Pediatrics. 2014;133(4):586-593.
- Kim K, Kim YH, Kim SY, et al. Low-dose abdominal CT for evaluating suspected appendicitis. N Engl J Med. 2012;366(17):1596-1605.
- Keyzer C, Tack D, de Maertelaer V, et al. Acute appendicitis: comparison of low-dose and standard-dose unenhanced multi-detector row CT. Radiology. 2004;232(1):164-172.
- Seo H, Lee KH, Kim HJ, et al. Diagnosis of acute appendicitis with sliding slab ray-sum interpretation of low-dose unenhanced CT and standard-dose i.v. contrast-enhanced CT scans. AJR Am J. Roentgenol. 2009;193(1):96-105.
- Ahn S. LOCAT (low-dose computed tomography for appendicitis trial) comparing clinical outcomes following low- vs standard-dose computed tomography as the first-line imaging test in adolescents and young adults with suspected acute appendicitis: study protocol for a randomized controlled trial. Trials. 2014;15:28.
- Alvarado A. A practical score for the early diagnosis of acute appendicitis. Ann Emerg Med. 1986;15(5):557-564.
- Ohle R, O’Reilly F, O’Brien KK, et al. The Alvarado score for predicting acute appendicitis: a systematic review. BMC medicine. 2011;9:139.
- McKay R, Shepherd J. The use of the clinical scoring system by Alvarado in the decision to perform computed tomography for acute appendicitis in the ED. Am J Emerg Med. 2007;25(5):489-493.
- Goldman RD, Carter S, Stephens D, et al. Prospective validation of the pediatric appendicitis score. J Pediatr. 2008;153(2):278-282.
- Wu HP, Chen CY, Kuo IT, et al. Diagnostic values of a single serum biomarker at different time points compared with Alvarado score and imaging examinations in pediatric appendicitis. J Surg Res. 2012;174(2):272-277.
- Calder JD, Gajraj H. Recent advances in the diagnosis and treatment of acute appendicitis. Br J Hosp Med. 1995;54(4):129-133.
- Agrawal CS, Adhikari S, Kumar M. Role of serum C-reactive protein and leukocyte count in the diagnosis of acute appendicitis in Nepalese population. NMCJ. 2008;10(1):11-15.
- Andersson RE, Hugander AP, Ghazi SH, et al. Diagnostic value of disease history, clinical presentation, and inflammatory parameters of appendicitis. World J. Surg. 1999;23(2):133-140.
- Keskek M, Tez M, Yoldas O, et al. Receiver operating characteristic analysis of leukocyte counts in operations for suspected appendicitis. Am J Emerg Med. 2008;26(7):769-772.
- Khan MN, Davie E, Irshad K. The role of white cell count and C-reactive protein in the diagnosis of acute appendicitis. JAMC. 2004;16(3):17-19.
- Mentes O, Eryilmaz M, Harlak A, et al. The value of serum fibrinogen level in the diagnosis of acute appendicitis. Ulus Travma Acil Cerrahi Derg. 2012;18(5):384-8.
- Ng KC, Lai SW. Clinical analysis of the related factors in acute appendicitis. Yale J Biol Med. 2002;75(1):41-45.
- Sengupta A, Bax G, Paterson-Brown S. White cell count and C-reactive protein measurement in patients with possible appendicitis. Ann R Coll Surg Engl. 2009;91(2):113-115.
- Xharra S, Gashi-Luci L, Xharra K, et al. Correlation of serum C-reactive protein, white blood count and neutrophil percentage with histopathology findings in acute appendicitis. World journal of emergency surgery: WJES. 2012;7(1):27.
- Yang HR, Wang YC, Chung PK, et al. Role of leukocyte count, neutrophil percentage, and C-reactive protein in the diagnosis of acute appendicitis in the elderly. Am Surg. 2005;71(4):344-347.
- Yu CW, Juan LI, Wu MH, et al. Systematic review and meta-analysis of the diagnostic accuracy of procalcitonin, C-reactive protein and white blood cell count for suspected acute appendicitis. Br J Surg. 2012.
- Al-Gaithy ZK. Clinical value of total white blood cells and neutrophil counts in patients with suspected appendicitis: retrospective study. World journal of emergency surgery : WJES. 2012;7(1):32.
- Cardall T, Glasser J, Guss DA. Clinical value of the total white blood cell count and temperature in the evaluation of patients with suspected appendicitis. Acad Emerg Med. 2004;11(10):1021-1027.
- Fergusson JA, Hitos K, Simpson E. Utility of white cell count and ultrasound in the diagnosis of acute appendicitis. ANZ J Surg. 2002;72(11):781-5.
- Kharbanda AB, Cosme Y, Liu K, et al. Discriminative accuracy of novel and traditional biomarkers in children with suspected appendicitis adjusted for duration of abdominal pain. Acad Emerg Med. 2011;18(6):567-574.
- Ortega-Deballon P, Ruiz de Adana-Belbel JC, Hernandez-Matias A, et al. Usefulness of laboratory data in the management of right iliac fossa pain in adults. Dis Colon Rectum. 2008;51(7):1093-1099.
- Yildirim O, Solak C, Kocer B, et al. The role of serum inflammatory markers in acute appendicitis and their success in preventing negative laparotomy. J Invest Surg. 2006;19(6):345-352.
- Paajanen H, Mansikka A, Laato M, et al. Novel serum inflammatory markers in acute appendicitis. Scand J Clin Lab Invest. 2002;62(8):579-584.
- Wu HP, Lin CY, Chang CF, et al. Predictive value of C-reactive protein at different cutoff levels in acute appendicitis. Am J Emerg Med. 2005;23(4):449-453.
- Vaughan-Shaw PG, Rees JR, Bell E, et al. Normal inflammatory markers in appendicitis: evidence from two independent cohort studies. JRSM Short Rep. 2011;2(5):43.
- Demircan M. Plasma d-lactate level: a useful marker to distinguish a perforated appendix from acute simple appendicitis. J Invest Surg. 2004 May-Jun;17(3):173-4;discussion 175.
- Yang HR, Wang YC, Chung PK, et al. Laboratory tests in patients with acute appendicitis. ANZ journal of surgery. 2006;76(1-2):71-74.
- Wang LT, Prentiss KA, Simon JZ, et al. The use of white blood cell count and left shift in the diagnosis of appendicitis in children. Pediatr Emerg Care. 2007;23(2):69-76.
- Andersson RE, Hugander A, Ravn H, et al. Repeated clinical and laboratory examinations in patients with an equivocal diagnosis of appendicitis. World J Surg. 2000;24(4):479-485.
- Andren-Sandberg A, Korner H. Quantitative and qualitative aspects of diagnosing acute appendicitis. Scand J Surg. 2004;93(1):4-9.
- Ebell MH. Diagnosis of appendicitis: part II. Laboratory and imaging tests. Am Fam Physician. 2008;77(8):1153-1155.
- Gronroos JM, Gronroos P. Leucocyte count and C-reactive protein in the diagnosis of acute appendicitis. Br J Surg. 1999;86(4):501-504.
- Gronroos P, Huhtinen H, Gronroos JM. Normal leukocyte count and C-reactive protein value do not effectively exclude acute appendicitis in children. Dis Colon Rectum. 2009;52(5):1028-1029.
- Stefanutti G, Ghirardo V, Gamba P. Inflammatory markers for acute appendicitis in children: are they helpful? J Pediatr Surg. 2007;42(5):773-776.
- van Dieijen-Visser MP, Go PM, Brombacher PJ. The value of laboratory tests in patients suspected of acute appendicitis. Eur J Clin Chem Clin Biochem. 1991;29(11):749-752.
- Murphy CG, Glickman JN, Tomczak K, et al. Acute appendicitis is characterized by a uniform and highly selective pattern of inflammatory gene expression. Mucosal Immunol. 2008;1(4):297-308.
- Rivera-Chavez FA, Peters-Hybki DL, Barber RC, et al. Innate immunity genes influence the severity of acute appendicitis. Ann Surg. 2004;240(2):269-277.
- Eriksson S, Granstrom L, Olander B, et al. Leucocyte elastase as a marker in the diagnosis of acute appendicitis. Eur J Surg. 1995;161(12):901-905.
- Sack U, Biereder B, Elouahidi T, et al. Diagnostic value of blood inflammatory markers for detection of acute appendicitis in children. BMC surgery. 2006;6:15.
- Lycopoulou L, Mamoulakis C, Hantzi E, et al. Serum amyloid A protein levels as a possible aid in the diagnosis of acute appendicitis in children. Clin Chem Lab Med. 2005;43(1):49-53.
- Muenzer JT, Jaffe DM, Schwulst SJ, et al. Evidence for a novel blood RNA diagnostic for pediatric appendicitis: the riboleukogram. Pediatr Emerg Care. 2010;26(5):333-338.
- Allister L, Bachur R, Glickman J, et al. Serum markers in acute appendicitis. J Surg Res. 2011;168(1):70-75.
- Kentsis A, Ahmed S, Kurek K, et al. Detection and diagnostic value of urine leucine-rich alpha-2-glycoprotein in children with suspected acute appendicitis. Ann Emerg Med. 2012;60(1):78-83 e71.
- Kentsis A, Lin YY, Kurek K, et al. Discovery and validation of urine markers of acute pediatric appendicitis using high-accuracy mass spectrometry. Ann Emerg Med. 2010;55(1):62-70 e64.
- Bealer JF, Colgin M. S100A8/A9: a potential new diagnostic aid for acute appendicitis. Acad Emerg Med. 2010;17(3):333-336.
- Mills AM, Huckins DS, Kwok H, et al. Diagnostic characteristics of S100A8/A9 in a multicenter study of patients with acute right lower quadrant abdominal pain. Acad Emerg Med. 2012;19(1):48-55.
- Noh H, Chang SJ, Han A. The diagnostic values of preoperative laboratory markers in children with complicated appendicitis. J Korean Surg Soc. 2012;83(4):237-41.