| Author | Affiliation |
|---|---|
| Jason Phan, MD | University of California Irvine, Department of Radiology, Orange, California |
| Chandana Lall, MD | University of California Irvine, Department of Radiology, Orange, California |
| Ross Moskowitz, MD | University of California Irvine, Department of Urology, Orange, California |
| Ralph Clayman, MD | University of California Irvine, Department of Urology, Orange, California |
| Jaime Landman, MD | University of California Irvine, Department of Urology, Orange, California |
ABSTRACT
Urinary tract interventions can lead to multiple complications in the renal collecting system, including retained foreign bodies from endourologic or percutaneous procedures, such as stents, nephrostomy tubes, and others. We report a case of very delayed erosion of embolization coils migrating into the renal pelvis, acting as a nidus for stone formation, causing mild obstruction and finally leading to gross hematuria roughly 18 years post transarterial embolization. History is significant for a remote unsuccessful endopyelotomy attempt that required an urgent embolization.
CASE REPORT
The patient is a 51-year-old Turkish man who recently presented to our institution with acute left flank pain and hematuria. Past history is significant for an atrophic nonfunctioning right kidney and a functioning left kidney with longstanding ureteropelvic junction stenosis. Since 1992, the patient has undergone an open dismembered pyeloplasty, 2 endopyelotomy attempts, and several stone treatments with ureteroscopy, holmium laser, and basket extraction.
During the second endopyelotomy attempt in 1992, a left renal subsegmental arterial branch was inadvertently traversed, resulting in acute hemorrhage and development of an arterial-ureteral fistula. This was embolized by interventional radiology with bare metal coils, with satisfactory hemostasis. A left ureteral stent was subsequently placed for the chronic ureteral obstruction.
After this procedure, the patient underwent routine ureteral stent changes every 2 years, which is a much longer period than that during which stents can typically be left in place before problems such as encrustation, infection, or obstruction are estimated to develop (2–4 months). Indwelling 7/14 French 28-cm endopyelotomy stents are used.
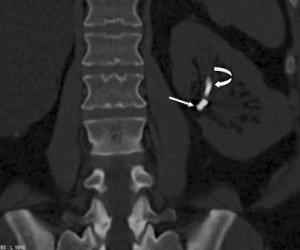
In 2010, the patient presented to our institution’s emergency department (ED) with significant hematuria and flank pain. Computed tomography (CT) of the abdomen and pelvis without intravenous contrast (stone protocol) was performed and showed several small oval and tubular high densities in the left renal pelvis, all of which were initially interpreted as renal calculi (Figure 1). The preexisting ureteral stent appeared intact and stable since the prior CT. The patient was then brought into the operating room for stent change and stone retrieval.
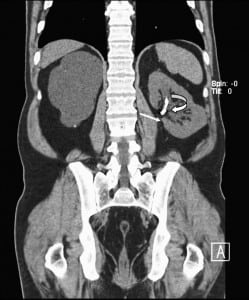
Intraoperatively, at least 2 bare metal coils were observed to endoscopically erode into the left renal pelvis. Few small renal calculi were also seen, most likely due to the coils acting as the nidus. Upon further review of the CT with multiple different windowing levels, the coils could be subtly distinguished from the stones owing to their tubular appearance and configuration of the tightly wrapped loops.
DISCUSSION
Patients with complex urologic histories present a unique challenge to the evaluating physician, be it an emergency department physician or a urologist assuming the care of a new patient. A thorough review of the surgical history, and identification of potential complications or morbidities, is essential in diagnosing the acute patient. This patient presented to the ED with complaints of left flank pain and hematuria, which in most situations would be fairly diagnostic of nephrolithiasis. This patient even had a CT showing a small density in the renal pelvis and a stent in place (Figure 1), supporting this diagnosis; however in a patient with this history, the differential is lengthened.
Any patient with gross hematuria should be evaluated for possible malignancy, such as urothelial or renal cell carcinoma (RCC). Stone disease, represented by densities on CT, can exist anywhere in the urinary collecting system, even within the renal parenchyma or embedded within the wall of the ureter, which can cause stricture. As previously mentioned, retained foreign materials from stents or nephrostomy tubes are also represented as densities on CT. These include fragments, strings, or the entire implant itself, and can be found with varying degrees of encrustation. In this case, embolization coils are the culprit (Figures 2 through 5).
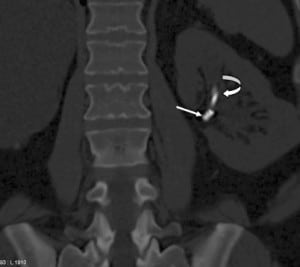
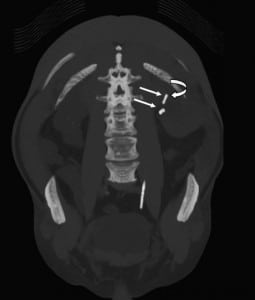
Procedures such as laser lithotripsy, endopyelotomy, and pyeloplasty all have inherent risks for vascular injury. Transarterial embolization is very effective in managing such vascular injury, since it has been widely used in the treatment of renal arteriovenous malformation (AVM), of acute extravasation in trauma patients, and for prophylaxis against operative blood loss before surgical resection of vascular tumors such as RCC and angiomyolipoma.
Complications have been widely reported for transarterial embolization during the procedure course as well as the immediate perioperative period.1–4 Commonly encountered complications are renal functional impairment and inadvertent embolizations of nontarget organs. Renal functional impairment often results from uremia, sepsis, and acute tubular necrosis owing to segmental parenchymal infarct.
Complications from inadvertent embolization of nontarget organ have been reported in the literature and include infarction and renal failure of the contralateral kidney; arteriovenous shunting within the targeted kidney, which can lead to embolization of the lungs and right-sided heart failure; adjacent bowel and skin necrosis; and thrombosis at the renal vein and inferior vena cava.5–7 With the newly designed coils, frequent complications encountered in the past, such as incomplete transcatheter expulsion of the coil and withdrawal into the aorta, have been almost completely eliminated.
Coil migration at the time of insertion happens commonly, but migration in the immediate perioperative period happens much less frequently. Patients undergoing prophylactic renal artery embolization within 24 hours before nephrectomy have had migrated coils at the time of nephrectomy.8 Delayed coil migration several years after the insertion is also very rare. Yoon et al9reported a case of migration of coils and guidewires from a treated renal AVM to the descending colon. Reed et al10 reported a case of passage of coil into the collecting system at 1 year post embolization.
Savoie et al11 reported a case in which the patient passed a stone containing a platinum coil. This event happened 5 years after percutaneous nephrolithotomy and embolization of a lower polar artery branch due to persistent hematuria. This coil was initially deployed too distally and had floated within the pseudoaneurysm cavity. Such encrustation of the coil with renal calculi is compatible with reports of migrated coils acting as the nidus for stone formation.12
In comparison to the other case reports, the erosion and migration of the coils in this patient happened at a much later time, about 18 years later. The patient was initially treated and followed up at an outside hospital in a different state for 10 years before transferring to our institution. This case underscores the need to remain vigilant for delayed coil migration beyond the intraoperative and immediate perioperative periods, and to search for the nidus of stone formation and unusual causes of obstruction as in this case.
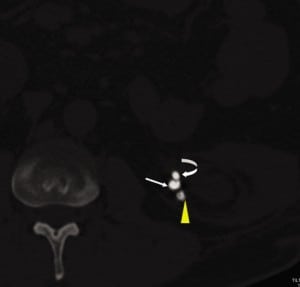
SUMMARY
Our report highlights the importance of checking for unusual causes of complications in patients who have undergone prior urologic intervention. The importance of viewing a stone protocol CT in different window settings cannot be overstated.
Footnotes
Supervising Section Editor: Rick A. McPheeters, DO
Submission history: Submitted May 2, 2011; Revision received June 21, 2011; Accepted July 19, 2011
Reprints available through open access at http://escholarship.org/uc/uciem_westjem
DOI: 10.5811/westjem.2011.7.6784
Address for Correspondence: Ross Moskowitz, MD
University of California Irvine, Department of Urology, 333 The City Blvd W, Ste 2100, Orange, CA 92868
E-mail: rmoskowi@uci.edu
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding, sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Chuang VP, Wallace S, Gianturco C, et al. Complications of coil embolization: prevention and management. AJR. 1981;137:809–881. [PubMed]
2. Klein FA, Texter JH, Mendez-Picon G. Complications of the Gianturco coil in preoperative infarction of renal cell carcinoma. J Urol. 1981;125:105–110. [PubMed]
3. Mazer MJ, Baltaxe HA, Wolf GL. Therapeutic embolization of the renal artery with Gianturco coils: limitations and technical pitfalls. Radiology. 1981;138:37–46. [PubMed]
4. Tisnado J, Beachley MC, Cho SR, et al. Peripheral embolization of a stainless steel coil. AJR.1979;133:324–326. [PubMed]
5. Lammer J, Justich E, Schreyer H, et al. Complications of renal tumor embolization. Cardiovasc Intervent Radiol. 1985;8:31–35. [PubMed]
6. Gralino BJ, Jr, Bicker DL. Staged endovascular occlusion of giant idiopathic renal arteriovenous fistula with platinum microcoils and silk suture threads. J Vasc Interv Radiol. 2002;13:747–752.[PubMed]
7. Wiesinger B, Schober W, Tepe G, et al. Complication after embolization of a complex renal vascular malformation. Nephrol Dial Transplant. 2005;20:1729–1733. [PubMed]
8. Wirthlin LS, Gross WS, James TP, et al. Renal artery occlusion from migration of stainless steel coils. JAMA. 1980;243:2064–2065. [PubMed]
9. Yoon JW, Koo JR, Baik GH, et al. Erosion of embolization coils and guidewires from the kidney to the colon: delayed complication from coil and guidewire occlusion of renal arteriovenous malformation. Am J Kidney Dis. 2004;43:1109–1112. [PubMed]
10. Reed A, Suri R, Marcovich R. Passage of embolization coil through urinary collecting system one year after embolization. Urology. 2007;70:1222.e17–1222.e18. [PubMed]
11. Savoie PH, Lafolie T, Gabaudan C, et al. Late complication of selective renal arterial embolization after percutaneous surgery: renal colic. Prog Urol. 2007;17:869–871. [PubMed]
12. Rutchik S, Wong P. Migration of arterial embolization coils as nidus for renal stone formation. J Urol. 2002;167:2520. [PubMed]


