| Author | Affiliation |
|---|---|
| Scott R. Samlan, MD | Resurrection Medical Center, Emergency Medicine Residency Program |
| Matthew T. Jordan, MD | Resurrection Medical Center, Emergency Medicine Residency Program |
| Shu B. Chan, MD, MS | Resurrection Medical Center, Emergency Medicine Residency Program |
| Michael S. Wahl, MD | Evanston Northwestern Healthcare, Illinois Poison Center |
| Rachel L. Rubin, MD | Resurrection Medical Center, Emergency Medicine Residency Program |
ABSTRACT
Introduction:
The development of tinnitus and/or hearing loss (THL) in patients receiving chronic salicylate therapy has been demonstrated. However, to date, little scientific data validates this relationship in the large single overdose setting. To correlate salicylate levels in patients with the subjective complaint of THL, following an acute salicylate overdose.
Methods:
A retrospective chart review of cases of acute salicylate toxicity and THL reported to the Illinois Poison Control Center (IPC) from 2001–2002 was performed. Data abstracted included age, gender, ingestion time, salicylate levels, and arterial blood gases.
Results:
Ninety-nine cases of THL were reviewed and analyzed with mean age of 23.7 years (SD: 10.9), 30.3% male, and 82.2% intentional overdoses. The average dose ingested was 20.0 grams (SD:20.2) and the mean time from ingestion to medical care was 12.4 hours (SD: 11.1). The mean initial ASA level was 48.3 mg/dl (SD: 16.4) with 86.9% having initial level ≥30mg/dl and 40.4% ≥50 mg/dl. 85.9% of cases presented to the hospital with their ASA level at or past peak. The mean pH was 7.45, pO2 = 108, pCO2 = 28.0, and HCO3 = 19.9.
Conclusion:
In this limited study, 85.9% of patients presenting with tinnitus and/or hearing loss following a single salicylate ingestion had initial salicylate levels at or past their peak and 86.9% were in the toxic range.
INTRODUCTION
Salicylates, aspirin in particular, have been used for their analgesic and anti-inflammatory properties for many years.1 Unfortunately, toxicity from salicylates remains a common occurrence in the emergency department (ED).2–4 Moreover, it is well known that the development of tinnitus is associated with salicylate use, and its onset has been heralded as an early marker of toxicity.5–7 This subjective complaint is classically described as a constant ringing, or roaring sound, perceived by the patient. Other presentations may include a subjective hearing loss or muffled hearing.8
Numerous reports have demonstrated the development of tinnitus and/or hearing loss (THL) in patients receiving chronic salicylate therapy.9–14 Anecdotally, its development has also been documented in the overdose setting.15 However, to date, little scientific data validates this relationship between THL and salicylate levels in patients with a large single salicylate overdose, as seen the ED. The hypothesis of this study was that the subjective complaint of tinnitus and/or hearing loss, in the setting of a large single salicylate overdose, correlates with an elevated salicylate level.
METHODS
A retrospective review of all cases of possible salicylate overdose reported to the Illinois Poison Center (IPC) from January 1, 2001 to December 31, 2002 was performed. Before the study’s inception, approval was obtained from the institutional review board of Resurrection Medical Center. Potential cases were identified by a search of the computerized database (dotlab) of the poison center. The search criteria included “aspirin” or “salicylate” entered in the search field based on the American Association of Poison Control Centers (AAPCC) coding of salicylate products. The database search had no restrictions for age, sex, or exclusions based on co-ingestants. The information recorded in the database was provided by the healthcare workers involved in the care of the patient through telephone consultation with the poison center. This information was then entered into the database by the employee of the poison center.
The study setting included all emergency departments, intensive care units, and urgent care centers in the state of Illinois who contacted the poison center regarding salicylate toxicity management. Treatment and management decisions were made by the healthcare provider and poison center specialist involved in each individual case.
The raw data abstracted from call records were reviewed individually for age, gender, timing from ingestion to presentation for treatment, salicylate levels, bicarbonate levels, pH level, pCO2 level, pO2 level, and presence or absence of THL. All names were de-identified and deleted from the database before data abstraction. These data elements were entered onto an Excel spreadsheet for analysis. An additional author to ensure entry accuracy reviewed approximately 10% of the collected data.
Since this was a retrospective chart review, the amount of detail provided in each chart varied and was not consistent for all charts. The time of ingestion to presentation for medical treatment was determined based on history taken by the healthcare provider and rounded off to the nearest half-hour. The presence of THL was also determined by the healthcare provider who queried this information during the initial contact with the patient. This measurement was performed utilizing a simple yes or no format. THL was defined by the poison center staff and medical provider as the subjective complaint of either ringing/roaring in the ears, “muffled” hearing, or the perception of a hearing loss by the patient
All poison center charts containing the search criteria words were reviewed for inclusion into the study. Reasons for exclusion from the study were as follows: patients that did not present to a health care facility after making poison center contact, patients who did not ingest a salicylate as demonstrated by two consecutive serum levels below 1 mg/dl, or did not have documented salicylate levels, patients who failed to have the presence or absence of tinnitus queried by the health care worker, patients who were unable to confirm the presence or absence of THL either due to age, language barrier, or alteration in consciousness.
Data Analysis
Following data collection, descriptive statistics were performed using Statistical Package for Social Sciences for Windows Version 11.5 (Chicago, IL, 2004).
RESULTS
The database search yielded follow-up records on 592 cases of salicylate toxicity. In 335 cases the presence or absence of THL was not queried during the encounter or its documentation was not provided. In 44 cases, the patient was unable to confirm the presence of THL due to either age, language barrier, or an alteration in their level of consciousness. Only 99 of the remaining 213 cases had documented THL and were included in the study. Table 1 gives the demographic profile of all 213 documented cases, while Figure 1 is a box-plot of the initial acetylsalicylic acid (ASA) level between patients with documented THL and patients with documented no THL.
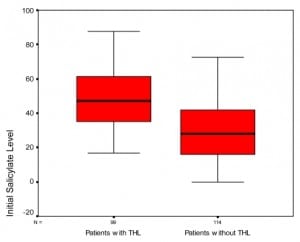
Boxplot of patients with and without tinnitus and/or hearing loss (THL) vs. initial aspirin levels.
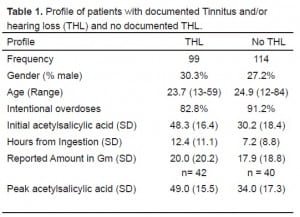
Profile of patients with documented Tinnitus and/or hearing loss (THL) and no documented THL.
There were minimal differences between THL and no THL patients in terms of demographics or cause of ASA toxicity. Patients with documented tinnitus or hearing loss had higher initial and peak ASA levels. For patients with THL, the mean initial ASA level was 48.3 mg/dl (SD: 16.4) compared to 30.2 mg/dl (SD:18.4) for patients without THL. Also, 86.9% of THL patients had initial level ≥30mg/dl and 40.4% had levels ≥50 mg/dl. On repeat testing, only 14.1% of cases had a further increase in ASA levels (mean increase = 8.5; SD: 4.6). Thus, 85.9% of cases presented to the hospital with their ASA level at or past peak. The average patient presented with compensated metabolic acidosis with mean pH = 7.45, mean pO2 = 108, mean pCO2 = 28.0, and mean HCO3 = 19.9.
DISCUSSION
The development of tinnitus and/or hearing loss (THL) has been heralded as an early marker of salicylate toxicity.5–7 However, this principle has only been demonstrated in patients receiving escalating doses of salicylates as a therapeutic measure.12,13 In the overdose setting, THL has been cited as a symptom, but no current studies document its true relationship to the ingestion. In our limited study, we provide support for this concept in the large overdose setting. The data presented from our sample demonstrates that those patients who develop THL have a 86% chance of having a salicylate level in the toxic range, or ≥30mg/dl.
The importance of this finding lies in the morbidity and mortality associated with salicylate toxicity. Intoxication can result in many deleterious effects on the human body, including death.16,17 As a result, the early recognition and treatment of salicylate toxicity remains paramount. In particular, prior studies have demonstrated that patients with critical serum salicylate levels have better outcomes when treatment is initiated earlier in their hospitalization.18,19
In our study we utilized THL as a subjective marker of toxicity. Our results demonstrate that the patient complaint of THL, following a single salicylate ingestion, is suggestive of a toxic salicylate level. The mean initial salicylate level in the THL patients was 48.3 mg/dl. It was also noted that 85% of the patients presented to the hospital with their salicylate level at their peak. This finding coincides with a prior study that documented that the two most common findings in patients with significant salicylate overdoses were the historical account of ingestion and the subjective complaint of tinnitus.20
The treatment of salicylate toxicity includes bowel decontamination, fluid resuscitation, urinary alkalinization, and extracorporeal elimination.8,21,22 Bowel decontamination in the form of activated charcoal is recommended for the majority of cases in salicylate overdose.2,21 However, beyond bowel decontamination, the consensus on other treatment modalities remains varied.2,3,23 In particular, recent publications have recommended the initiation of urinary alkalinization when the serum salicylate level is found above 35 mg/dl.8,21
As stated earlier, the measured serum salicylate level may take a significant time to obtain. 24 Consequently, flowcharts have been developed utilizing the patient’s symptoms or other rapidly available clinical data to help guide treatment decisions.7,22 Most of these diagrams list THL as a marker of early toxicity typically presenting at a level of 25 mg/dl.5,8 The data from our study shows that 86.9% of patients had a level ≥30mg/dl.
The principle that THL typically occurs around 25 mg/dl is based on previous studies over the past five decades.9–14 Mongan, et al13 demonstrated that THL was observed at an average salicylate concentration of 29.5 mg/dl in patients receiving chronic salicylate therapy. A study by Myers10 reported sensorineural hearing loss as the plasma salicylate level approached 30 mg/dl. As stated previously, both these studies utilized escalating doses of salicylates as a therapeutic, and both are distinctly different than the typical ED overdose setting. In our study, the mean salicylate level for those with THL was 48.3 mg/dl, much higher than this previously stated level of 25 mg/dl.
LIMITATIONS
Our study possesses several limitations. First, the self reporting of THL by the patient creates some potential problems. No accurate method was available to determine the validity of the patient’s account. It is possible that some patients may have falsely denied or affirmed the presence of THL. Next, since this was a retrospective chart review, the amount of detail provided in each chart varied. Moreover, no ED or inpatient charts were obtained in this study to confirm poison center records. Lab values and patient histories were strictly based on the treating healthcare provider’s phone testimony. In addition, the type of salicylate, and its form (i.e. enteric coated), were both sparsely documented making any analysis of these factors difficult. There was a limited population in our sample that had taken co-ingestants as well as prescribed medicine in conjunction with their salicylate ingestions. In reviewing the data, it seems however, that there were no known co-ingestants that would have contributed to tinnitus, but we acknowledge that this is a limitation in this study. The provided treatment modalities were also not always identical. Their technique and timing unfortunately could have affected a patient’s symptoms and lab values. The data also shows that the patients with tinnitus presented later in their course (12.4 versus 7.2 hours). But on the same note, 85.9% of the cases had the initial level above or at their peak salicylate level and the difference between initial and peak levels were very small in each group. Therefore we do not believe that the time presentation difference to be a large confounding variable. Finally, our study is limited in the fact that from a large initial study sample, only 213 had documentation of tinnitus or hearing loss. The lack of documentation of 335 patients prevents statistical testing between the groups. It may be that there were a number of patients without THL but with elevated salicylate levels. Thus, it is impossible to determine at what cutoff salicylate level we would expect THL but the evidence in this study does show that if THL occurs, we can expect an elevated salicylate level.
CONCLUSION
In the emergency department setting, it is essential to consider the diagnosis of salicylate ingestion when an overdose is suspected and a patient complains of THL. This limited study demonstrates that 85.6% of patients with single large salicylate ingestion combined with the complaint of tinnitus, and/or hearing loss, had initial salicylate level at or past their peak and 86.9% were in the toxic range.
Footnotes
Supervising Section Editor: Brandon K. Wills, DO, MS
Submission history: Submitted November 20, 2007; Revision Received March 10, 2008; Accepted March 14, 2008
Full text available through open access at http://escholarship.org/uc/uciem_westjem
Address for Correspondence: Shu Chan, MD, MS, Resurrection Medical Center, Emergency Medicine Residency Program, 7435 West Talcott Avenue, Chicago, Illinois 60631
E-mail: schan@reshealthcare.org
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Boettcher FA, Salvi RJ. Salicylate ototoxicity: review and synthesis. Am J Otolaryngol.1991;12:33–47. [PubMed]
2. Yip L, Dart RC, Gabow PA. Concepts and controversies in salicylate toxicity. Emerg Med Clin North Am. 1994;12:351–364. [PubMed]
3. Proudfoot AT. Toxicity of salicylates. Am J Med. 1983;75:99–103. [PubMed]
4. Patel DK, Hesse A, Ogunbona A, Notarianni LJ, Bennett PN. Metabolism of aspirin after therapeutic and toxic doses. Hum Exp Toxicol. 1990;9:131–136. [PubMed]
5. Dargan PI, Wallace CI, Jones AL. An evidence based flowchart to guide the management of acute salicylate (aspirin) overdose. Emerg Med J. 2002;19:206–209.[PMC free article] [PubMed]
6. Temple AR. Pathophysiology of aspirin overdosage toxicity, with implications for management. Pediatrics. 1978;62:873–876. [PubMed]
7. Krause DS, Wolf BA, Shaw LM. Acute aspirin overdose: mechanisms of toxicity. Ther Drug Monit. 1992;14:441–451. [PubMed]
8. Donovan JW, Akhtar J. Salicylates. In: Ford M, Delany K, Ling L, Erickson T, editors.Clinical Toxicology. Philadelphia: W.B. Sanders; 2001. pp. 275–280.
9. Jardini L, Findlay R, Burgi E, Hinderer K, Agarwal A. Auditory changes associated with moderate blood salicylate levels. Rheumatol Rehabil. 1978;17:233–236. [PubMed]
10. Myers EN, Bernstein JM, Fostiropolous G. Salicylate ototoxicity: a clinical study. N Engl J Med. 1965;273:587–90. [PubMed]
11. Halla JT, Atchison SL, Hardin JG. Symptomatic salicylate ototoxicity: a useful indicator of serum salicylate concentration? Ann Rheum Dis. 1991;50:682–684.[PMC free article] [PubMed]
12. Halla JT, Hardin JG. Salicylate ototoxicity in patients with rheumatoid arthritis: a controlled study. Ann Rheum Dis. 1988;47:134–137. [PMC free article] [PubMed]
13. Mongan E, Kelly P, Nies K, Porter WW, Paulus HE. Tinnitus as an Indication of Therapeutic Serum Salicylate Levels. JAMA. 1973;226:142–145. [PubMed]
14. McCabe PA, Dey FL. The effect of aspirin upon auditory sensitivity. Ann Otol Rhinol Laryngol. 1965;74:312–325. [PubMed]
15. Beveridge GW, Forshall W, Munro JF, Owen JA, Weston IA. Acute Salicylate Poisoning in Adults. Lancet. 1964;41:1406–1409. [PubMed]
16. Strom J, Thisted B, Krantz T, Bredgaard SM. Self-poisoning treated in an ICU: drug pattern, acute mortality and short-term survival. Acta Anaesthesiol Scand.1986;30:148–153. [PubMed]
17. Chapman BJ, Proudfoot AT. Adult salicylate poisoning: deaths and outcome in patients with high plasma salicylate concentrations. Q J Med. 1989;72:699–707.[PubMed]
18. Thisted B, Krantz T, Stroom J, Sorensen MB. Acute salicylate self-poisoning in 177 consecutive patients treated in ICU. Acta Anaesthesiol Scand. 1987;31:312–316.[PubMed]
19. Anderson RJ, Potts DE, Gabow PA, Rumack BH, Schrier RW. Unrecognized adult salicylate intoxication. Ann Intern Med. 1976;85:745–748. [PubMed]
20. Jammehdiabadi M, Tierney M. Impact of toxicology screens in the diagnosis of a suspected overdose: salicylates, tricyclic antidepressants, and benzodiazepines. Vet Hum Toxicol. 1991;33:40–43. [PubMed]
21. Flomenbaum NE. Salicylates. In: Goldfrank LR, Flomenbaum NE, Lewin NA, Howland MA, Hoffman RJ, Nelson LS, editors. Goldfrank’s Toxologic Emergencies. New York: McGraw-Hill; 2002. pp. 507–518.
22. Notarianni L. A reassessment of the treatment of salicylate poisoning. Drug Saf.1992;7:292–303. [PubMed]
23. Prescott LF, Balali-Mood M, Critchley JA, Johnstone AF, Proudfoot AT. Diuresis or urinary alkalinisation for salicylate poisoning? Br Med J (Clin Res Ed) 1982;285:1383–1386.
24. Song W, Dou C. One-step immunoassay for acetaminophen and salicylate in serum, plasma, and whole blood. J Anal Toxicol. 2003;27:366–371. [PubMed]


