| Author | Affiliation |
|---|---|
| Sanjay Arora, MD | University of Southern California, Keck School of Medicine, Los Angeles, CA |
| Theodore Long | University of Southern California, Keck School of Medicine, Los Angeles, CA |
| Michael Menchine, MD, MPH | University of Southern California, Keck School of Medicine, Los Angeles, CA |
ABSTRACT
Introduction:
To evaluate the test characteristics of the urine dipstick as a screening tool for elevated serum creatinine in patients with uncontrolled diabetes mellitus in the emergency department (ED).
Methods:
Patients with diabetes over the age of 18 who presented to the ED for any complaint over a three-month study period were considered eligible for participation in this study. A finger-stick blood glucose of ≥250 mg/dL at triage was used to confirm the diagnosis of uncontrolled diabetes. After obtaining written consent, each patient had a urine dip performed and a chemistry panel drawn. Any level of proteinuria on the urine dip was considered to be a positive test. Based on the laboratory and clinical guidelines at our institution, renal insufficiency was defined as creatinine concentration of greater than 1.3 mg/dL.
Results:
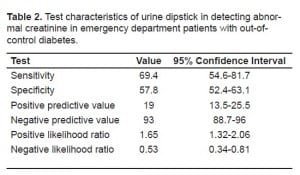
Three Hundred ninety-three confirmed patients with uncontrolled diabetes were enrolled in this study, and 49 of these (12.5%) were found to have renal insufficiency. The sensitivity and specificity of the urine dip for predicting renal insufficiency were 69.4% (95% confidence interval [CI] 54.6–81.7%) and 57.8% (95%CI 52.4–63.1%) respectively. The positive predictive value was 19% (95%CI 13.5–25.5%), and the negative predictive value was 93% (95%CI 88.7–96%). The positive likelihood ratio was 1.65 (95%CI 1.32–2.06) and the negative likelihood ratio was 0.53 (95%CI 0.34–0.81).
Conclusion:
In this cohort of patients with uncontrolled diabetes, the test characteristics of the urine dipstick make it a poor screening tool for renal insufficiency in the ED.
INTRODUCTION
Diabetes mellitus is the foremost cause of end-stage renal disease in the United States.1 Diabetes-specific changes in blood flow in the kidney stemming from glomerulosclerosis put poorly controlled patients with diabetes at high risk for clinically significant nephropathy.2 As such, the threshold to screen patients who present in the emergency department (ED) with uncontrolled diabetes for renal dysfunction has always been low. In addition, the added fear of contrast-induced nephropathy has led to routine serum creatinine screening in most patients with diabetes prior to contrast enhanced computed tomography.3 As the prevalence of patients with diabetes seen in the ED continues to rise, a rapid way to screen for renal dysfunction becomes increasingly important.
The urine dipstick is commonly used in EDs to look for white blood cells and other markers of urinary tract infections; to check for red blood cells in patients with suspected renal stones; and to detect ketones in patients with diabetes who are suspected of being in diabetic ketoacidosis (DKA). The dipstick test has the advantages of being inexpensive, quick and noninvasive. Because of these advantages, investigators have examined the urine dipstick’s usefulness as a screening tool for renal insufficiency with hopes of obviating the need for serum creatinine testing. The urine dipstick has previously been evaluated as a routine screening test for both renal and cardiovascular disease in an outpatient setting.4,5 These studies of outpatients with and without risk factors for renal insufficiency have yielded generally poor results. In contrast, studies that included only specific, high-risk patient populations, such as hypertensive patients, have shown much more promise for expanding the role of the urine dipstick as a screening tool for renal insufficiency.6 Therefore, we sought to evaluate the test characteristics of the urine dipstick as a screening tool for elevated serum creatinine in patients with known or newly diagnosed diabetes and random blood sugar > 250 mg/dl.
METHODS
The institutional review board of the Los Angeles County Hospital at University of Southern California (LAC+USC) approved this prospective study, and we obtained written informed consent from all study participants. The cohort studied represents a convenience sample of ED patients presenting between the hours of 9am and 5pm from October 2007 to December 2007. All adult patients with known or suspected diabetes who presented to ED triage for any reason were screened for enrollment. Obtaining a blood glucose value at triage was at the discretion of the triage nurse and was not a formal part of the study protocol. Only those patients with blood glucose at triage of at least 250 mg/dL were considered eligible for enrollment. This value was chosen as it is a generally accepted tipping point endorsed by the American Diabetes Association at which many triage protocols and practicing emergency physicians initiate a laboratory workup looking for evidence of acute and chronic ailments (i.e. renal dysfunction, DKA, infection).7 Subjects were excluded if they were judged as not being in a mental and/or physical state to provide informed consent.
All enrolled patients had both a urine dipstick (Bayer multistix 10SG) performed to look for protein, and a blood draw to assess serum creatinine. Any level of proteinuria on the urine dip was considered to be a positive test, and per the manufacturer the cutoff value is 20 mg/L. We did not record the exact level of proteinuria. Renal insufficiency was defined as serum creatinine concentration of greater than 1.3 mg/dL. This value is based on the laboratory and clinical guidelines at our institution for renal dysfunction. The urine dipstick was read by the treating nurse and relayed to a research assistant in real time. We analyzed data using STATA version 9.0. Sensitivity, specificity, predictive values, likelihood ratios and corresponding confidence intervals were determined for the urine dip using the ‘diagt’ command.
RESULTS
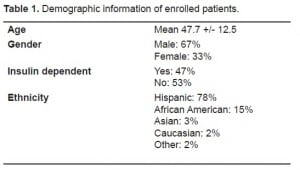
Of 513 patients with uncontrolled diabetes identified in ED triage in the three-month study period, 393 agreed to participate. Characteristics of the sample are given in Table 1. Forty-nine (12.5%) had renal insufficiency defined as a serum creatinine level of greater than 1.3 mg/dL. The urine dip was positive for proteinuria in 179 (45.5%) patients. The urine dipstick was 69.4% sensitive (95% confidence interval [CI], 54.6–81.7%) and 57.8% specific (95% CI, 52.4–63.1%) in this sample (Table 2). The positive predictive value of the urine dip was 19% (95% CI, 13.5–25.5%) and the negative predictive value was 93% (95% CI, 88.7–96%). The positive likelihood ratio was 1.65 (95% CI, 1.32–2.06) and the negative likelihood ratio was 0.53 (95% CI, 0.34–0.81).
DISCUSSION
The urine dipstick is a rapid, noninvasive and inexpensive means of screening patients during ED triage for a variety of potential disease states. This has prompted clinician scientists to attempt to expand its usefulness by studying its ability to screen for renal insufficiency. Although results from prior studies are varied, some believe it is possible that the urine dip may be more reliable if used as a screening tool for renal dysfunction in a more specific and well-defined patient population.6
Glauser et al8 performed a retrospective study examining whether urine protein could replace serum creatinine as a rapid screening test for renal dysfunction in patients considered to be at low risk for renal disease. They defined their low risk cohort by the absence of hypertension, diabetes mellitus, multiple myeloma and systemic lupus erythematosus. They retrospectively identified 310 patients who met their low-risk criteria and had a serum creatinine level and negative urine protein assessment based on chart review. They found that none of the patients in their cohort had an elevated creatinine level. This study was limited by a high cutoff for renal insufficiency (greater than 2.0 mg/dL) and by the fact that not all patients with a negative urine protein truly received the gold standard test for renal dysfunction.
Firestone et al9 conducted a retrospective chart review examining the utility of urine dipstick as a screening tool in ED patients. Their goal was to identify a set of risk factors whose absence, combined with a negative urine dip, could rule out renal dysfunction. After a review of over 2400 charts, they concluded that in isolation, the sensitivity and negative predictive value of the urine dip in detecting renal insufficiency were 85.5% and 96.2% respectively. All patients with elevated creatinine were found to have at least one of the following high-risk conditions: prior renal disease, hypertension, diabetes, congestive heart failure or age >60. The authors conclude that the sensitivity of the urine dip was inadequate to merit its use as a stand-alone test, but if combined with a clinical questionnaire adequate sensitivity may be reached.
Subsequently, Firestone et al10 prospectively collected data on 1,354 ED patients, including urine dipstick results, triage vital signs, basic demographic data, 14 medical history items and serum creatinine results. Through logistic regression they identified the following predictor variables associated with elevated creatinine: age >60 years, known renal insufficiency, diabetes, hypertension, diuretic use, vomiting, and proteinuria. They concluded a negative urine dipstick combined with the absence of the aforementioned risk factors result in a dipstick sensitivity of 98.1% (95.8–99.9%) and a specificity of 21.2% (18.8–23.2%).
Given the success of the urine dip in screening for renal insufficiency in a very well-defined, low-risk group of patients, we sought to determine if it could be used to screen for renal insufficiency in a well-defined, high-risk group of patients. After considering all potential predictor variables and chronic diseases, we chose to examine the utility of the urine dipstick for detecting elevated creatinine levels in ED patients with uncontrolled diabetes. We chose this group based on the high prevalence of disease among ED patients, as well as the severity of disease specific and iatrogenic (i.e. contrast-induced nephropathy) renal dysfunction in this group. Unfortunately, our results show that in this high-risk group of ED patients with uncontrolled diabetes, the poor calculated test characteristics do not make the urine dipstick a viable screening tool for elevated creatinine levels.
LIMITATIONS
When designing this prospective trial we worked to minimize bias and other sources of potential error; however, some limitations warrant mentioning. First, the interpretation of dipstick results is somewhat subjective, and as all nurses have their clinical laboratory improvement amendments (CLIA) certification, we did not offer additional training to the nurses who interpreted the urine dips, nor did we have multiple staff members read the same dipstick to ensure that we had adequate inter-rater reliability. That being said, the health professionals responsible for interpreting and recording the urine dip results are already very familiar and comfortable with this process and do this daily as part of their daily clinical care. Also, subjectivity exists in assigning an exact level to a positive test and significantly less existing in purely distinguishing positive from negative. The staff reading the dipsticks was not blinded to the purpose of the study, but this too was unnecessary as the creatinine value was not obtained until a later point in each patient’s ED workup.
Second, the glomerular filtration rate, and not an elevated serum creatinine, is truly the gold standard for renal dysfunction. We chose to use serum creatinine as a surrogate for renal function in our study because in the ED this is the measurement used in clinical decision-making, and it is much easier to obtain and more commonly used in prior studies on the same topic. As such, although imperfect, the serum creatinine seems to be the most appropriate reference standard for our purposes. We defined renal dysfunction as a serum creatinine concentration of greater than 1.3 mg/dL. Other published studies have used differing values (i.g. 1.5 or 2.0 mg/dL) based on what the laboratories at their specific institutions consider to be abnormal. Our value was chosen after a discussion with the clinical laboratory at our hospital. If we had used either a value of 1.5 or 2.0 mg/dl the sensitivity and specificity still do not reach acceptable values. (Sensitivities 67.6%, 95%CI 50.2–82% and 71.4%, 95%CI 41.9–91.6% respectively; specificities 56.7%, 95%CI 51.4–62% and 55.4%, 95%CI 50.2–60.5% respectively).
Third, we did not screen the patients in our study for co-occurring diseases that might have affected the urine dipstick results. We knew the enrolled patients were at high risk for renal disease based on their uncontrolled diabetes, which could affect their level of proteinuria. Had we also recorded the presence or absence of other high-risk diseases identified in previous studies we could have attempted to evaluate test characteristics of the urine dipstick in combination with other high-risk disease states (i.e. patients with diabetes and hypertension). However, it seemed to be reasonable to start with just one condition. Finally, this study was conducted at a single urban ED with a largely Latino patient population and thus has limited external generalizibility.
CONCLUSION
Our study contributes to the growing knowledge base concerning the use of the urine dip by offering data from the specific high-risk population of patients with diabetes. We found in our cohort of patients with uncontrolled diabetes, the urine dipstick had a sensitivity of 68.6% (95% CI, 49.9–83.2%) and a specificity of 55.2% (95% CI, 48.4–62.0%) in screening for renal insufficiency. Our results suggest that dipstick analysis should not be used as a stand-alone screening test for renal insufficiency and cannot be used as a replacement for obtaining a serum creatinine in patients with diabetes. Further study is warranted to confirm these findings in a broader array of clinical settings.
Footnotes
Supervising Section Editor: Laleh Gharahbaghian, MD
Submission history: Submitted February 23, 2010; Revision received April 22, 2010; Accepted May 7, 2010
Reprints available through open access at http://escholarship.org/uc/uciem_westjem.
Address for Correspondence: Sanjay Arora, MD, 1200 North State Street, Room 1011, Los Angeles, CA 90033
Email: sanjay.arora@usc.edu
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. US Renal Data System, USRDS 2009 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States. National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; Bethesda, MD: 2009.
2. Sego S. Pathophysiology of diabetic nephropathy. Nephrol Nurs J. 2007;34(6):631–3. [PubMed]
3. Parfrey P. The clinical epidemiology of contrast-induced nephropathy. Cardiovasc Intervent Radiol. 2005;S2:S3–11. 28. Review. [PubMed]
4. de Jong PE, Gansevoort RT, Bakker SJ. Macroalbuminuria and microalbuminuria: do both predict renal and cardiovascular events with similar strength? J Nephrol. 2007 Jul-Aug;20(4):375–80.[PubMed]
5. Madison JR, Spies C, Schatz IJ, et al. Proteinuria and risk for stroke and coronary heart disease during 27 years of follow-up: the Honolulu Heart Program. Arch Intern Med. 2006 Apr;166(8):884–9. [PubMed]
6. Agarwal R, Panesar A, Lewis RR. Dipstick proteinuria: can it guide hypertension management? Am J Kidney Dis. 2002 Jun;39(6):1190–5. [PubMed]
7. American Diabetes Association From Diabetes Care. 2006;29(12)
8. Glauser J, Montgomery A. Urine protein as a rapid screen for renal function in the ED: can it replace serum creatinine in selected patients? Emerg Radiol. 2004 Jul;10(6):319–22. [PubMed]
9. Firestone D, Wos A, Killeen JP, et al. Can urine dipstick be used as a surrogate for serum creatinine in emergency department patients who undergo contrast studies? J Emerg Med. 2007 Aug;33(2):119–22. [PubMed]
10. Firestone D, Band R, Hollander J, et al. Use of a urine dipstick and brief clinical questionnaire to predict an abnormal serum creatinine in the emergency department. Acad Emerg Med. 2009 Aug;16(8):699–703. [PubMed]


