| Author | Affiliation |
|---|---|
| Amy A. Ernst, MD | University of New Mexico, Department of Emergency Medicine, Albuquerque, New Mexico |
| William A. McIntyre, MD | University of New Mexico, Department of Emergency Medicine, Albuquerque, New Mexico |
| Steven J. Weiss, MD | University of New Mexico, Department of Emergency Medicine, Albuquerque, New Mexico |
| Chad Berryman | University of New Mexico, Department of Emergency Medicine, Albuquerque, New Mexico |
ABSTRACT
Introduction:
Many traumatic pneumothoraces (PTX) are not seen on initial chest radiograph (CR) (occult PTX) but are detected only on computed tomography (CT). Although CR remains the first tool for detecting PTX, most trauma patients with significant thoracoabdominal injuries will receive both CT and CR. The primary objective of this study was to retrospectively determine the effectiveness of CR for detecting PTX in trauma patients. Our hypotheses were that CR is a sensitive indicator of PTX on CT, that chest pain and shortness of breath are good predictors of PTX on CR, and that we could determine other predictors of PTX on CR.
Methods:
All trauma patients presenting to our Level I trauma center with a CT-diagnosed PTX over a 2-year period who had both a CR and a chest CT were included. The CT reading was considered the gold standard for PTX diagnosis. Electronic medical records were searched using key words for diagnoses, symptoms, demographics, and radiologic results. We recorded the official radiologist readings for both CR and CT (positive or negative) and the size of the PTX on CT (large, moderate, small, or tiny). The outcome variable was dichotomized based on presence or absence of PTX detected on CR. Descriptive statistics and χ2 tests were used for univariate analysis. A regression analysis was performed to determine characteristics predictive of a PTX on CR, and 1 variable was added to the model for every 10 positive CRs. With equal-size groups, this study has the power of 80% to detect a 10% absolute difference in single predictors of PTX on CR with 45 subjects in each group.
Results:
There were 134 CT-documented PTXs included in the study. Mean age was 42, and 74% were men. For 66 (49%) patients, PTX was detected on CR (sensitivity = 50%). The CR detected 30% of small PTX, 35% of moderate PTX, and 33% of large PTX. Comparing patients with and without PTX on CR, there were no significant differences in shortness of breath or chest pain. There no relationships between PTX detected on CR and age, gender, penetrating versus blunt injury, bilaterality of the PTX, or presence of lung contusion or hemothorax on CT. After adjusting for all significant variables, predictor of a PTX detected on CR was air in the tissue on CR (adjusted odds ratio [OR] = 3.8) and PTX size (compared to a tiny PTX, adjusted OR = 2.0 for a small PTX, 7.5 for a moderate PTX, and 51 for a large PTX). Chest tubes were used in 89% of patients with PTX on CR and 44% of patients with PTX only on CT (difference 45%; 95% confidence interval 30, 58).
Conclusion:
Factors associated with PTX on CR included air in the soft tissue on CR and size of the PTX. Even when PTX is not apparent on CR, 44% of these PTXs received placement of a chest tube.
INTRODUCTION
More than 50,000 traumatic pneumothoraces (PTX) occur in the United States annually; PTX is the second most common traumatic chest injury, and it is seen in 40% to 50% of patients with chest trauma.1,2 Occult PTX (OPTX), that is, PTXs not detected by clinical examination or a chest radiograph (CR) but later diagnosed by computed tomography (CT), have been shown to occur in 54.8% of chest trauma cases.3 A small undetected PTX can rapidly progress to tension PTX, severe dyspnea, and hemodynamic collapse,2,4thereby increasing mortality rates in trauma patients.5 This is particularly important in patients receiving positive pressure ventilation, where it has been demonstrated that 38% of OPTX progress.4 Early detection is essential and could influence ongoing assessment and management1,6 and could possibly be a life-saving intervention before a CT can be safely performed.
Trauma patients in the emergency department (ED) are necessarily supine, and air in the pleural space moves anteromedially; thus, the pleural line typically used to diagnose PTX in an upright anteroposterior (AP) CR is not generally seen. It is well established that many traumatic PTXs are not seen on an AP supine CR (SCR).1 However, currently the AP SCR remains the first diagnostic tool for the detection of PTX. With the increased use of abdominal and thoracic CT, however, there has been a predictable increase in the published incidence of OPTX diagnoses by CT ranging from 3.7% in injured children and to 64% in intubated patients with multiple trauma.3,7–9 The incidence numbers reflect the type of trauma patient.
Although Wolfman et al1 provided a size classification system and management approach based on the CT, there is still considerable debate whether this changes management of a patient with OPTX because many can be treated expectantly.8,10 Most importantly, there is criticism regarding the excessive use of CT because of their minimal impact in changing the management of patients with OPTX; some argue for the refinement of CT use to identify critical lesions instead of trivial findings.11 Although CT will continue to be the gold standard for diagnosing PTX, there is scant evidence in the literature accurately characterizing the ability of AP SCR to detect PTX in trauma patients. The vast majority of studies simply report the increased incidence of OPTX with the advent of CT without providing any statistical analysis comparing AP SCR to CT. Furthermore, the studies include patients from select categories, such as “thoracic trauma” and “chest or multiple trauma,” or ill-defined categories, such as “blunt trauma,” without specifically describing which patients qualify, which makes comparisons and application difficult or impossible. Recent studies comparing thoracic ultrasound to CR and CT in the emergency setting have reported an extreme variability in the sensitivity of AP SCR for detecting PTX ranging from 27% to 75% when using CT as the gold standard.12–14 Accurately characterizing the sensitivity of AP SCR, especially for particular subsets of patients, may help clinicians determine which patients need to be more closely monitored and potentially which less severe patients need only be monitored without having to undergo a CT at all.
In our setting, patients with chest pain after trauma, very major trauma, and/or altered mental status with a mechanism that could result in chest trauma (eg, abrasions on the chest, ecchymoses) generally had a CT chest and CR performed. This is based on severity and mechanism and subject to the discretion of the physician.
The primary objective of this study was to determine the effectives of CR for detecting PTX in trauma patients. Our hypotheses were that CR is a sensitive indicator of PTX on CT, that chest pain and shortness of breath are good predictors of PTX on CR, and that we could determine other predictors of PTX on CR.
METHODS
Study Design and Setting
This was a retrospective cohort study at an urban ED with an emergency medicine residency program with approximately 75,000 patient visits per year. There is a trauma team onsite and major trauma patients undergo a trauma activation protocol (TAP) based on severity of injuries and mechanisms of injury. The site is the only Level I trauma center in the state.
At our institution activation of the trauma team is based on physiologic abnormalities and mechanism of injury. Vital signs, severity, of injury and expected time of arrival are paged to the team, which includes a trauma attending physician, an ED attending physician, residents, radiology technicians, respiratory therapists, and a pharmacist. The TAP depends on multiple possible factors, including airway, breathing, or circulation compromise; disability (neurologic deterioration); exposure (penetrating injuries, amputations, crush injuries, burns); and pregnancy with significant mechanism. Mechanisms such as rollover, ejection, and prolonged extrication time are also included in the TAP mechanism.
A focused assessment with sonography for trauma (FAST) exam is typically included in the trauma room, especially for patients with major traumas and any alteration of level of conscious and depending on mechanism of injury. It is up to the discretion of the team at the time of injury. The lungs are not typically included in the evaluation at our institution.
An electronic trauma database is kept updated with all trauma patients who are seen and/or admitted by the trauma service. The ED or trauma physicians order all imaging while the patient is in the trauma area. This is done either verbally or on a separate paper order form, which is then entered by a technician. Almost universally a patient with a chest injury in the major trauma area undergoes initial plain films of the chest and abdomen, then proceeds to the CT scanner for a CT of the chest and/or other areas. However, during the period of the study there was no requirement for imaging.
For research purposes the researchers obtained access to the trauma database. The study was approved as exempt by the institution’s Human Research Review Committee.
Selection of Participants
All consecutive trauma patients presenting to our Level I trauma center with a CT-diagnosed PTX over a 2-year period (2008–2010) who had both a CR and a chest CT were included. The CT reading was considered the gold standard for PTX diagnosis. Electronic medical records were then searched for diagnoses, demographic data, and CT and CR results. Official readings from the radiologist were used.
Patients with chest trauma who did not have a CT of the chest and those who did not undergo an initial CR in the ED were excluded. Patients without a PTX were also excluded.
Each patient’s ED and hospital records were reviewed. Electronic medical records were searched for diagnoses, symptoms, demographics, and radiologic results. Key search words and terms were those used by the trauma data-entry personnel, including pneumothorax, lung collapse, chest trauma, chest tube, and lung injury; common abbreviations and abbreviations known to be used by data-entry personnel were also included. We recorded the official radiologist readings for both CR and CT (positive or negative) and the size of the PTX on CT (large, moderate, small, or tiny).
Data collected included demographics, medical history, CR reading, CT reading, size of PTX, whether surgery was needed, admission data, and whether a chest tube was placed. Additionally rib fractures (numbers), breath sounds (decreased), and presence of subcutaneous (SQ) air were recorded.
At our institution, chest tubes are inserted at the discretion of the physician. Typical indicators include PTX with significant size (ie, too large for a pigtail catheter) and significant clinical symptoms, PTX with potential for increase in size, or a hemothorax or other complicating issues.
Training for Data Gathering and Agreement
A data collection sheet was formulated from several sessions over a period of approximately 2 months based on previous studies in the literature. Data were obtained by 2 trained abstractors. Meetings and sessions to educate the abstractors on data collection and methods of analysis were held over a period of approximately 2 weeks. In addition, the data gathered were analyzed by another author of the study to determine agreement. A κ statistic was calculated to measure agreement in observations between authors in all data collection for approximately 20% of the data. This was chosen as a representative of the sampling and thought to be adequate to determine agreement.
PTX Size Determination
Our radiologists use a well-defined approach to categorizing the size of PTX. The tiny category was defined as less than 1 cm thick on the CT.15 The small category was defined as <2 cm presence of a visible rim or approximately 10% PTX. The medium category included all cases of PTX between the small and large parameters. A large PTX was defined as 31% or greater. This is based on previous literature calculating the size of the PTX based on the size of the space between the deflated lung and the chest wall. In patients with large PTX, the size of the space is more than 2 cm, which corresponds to greater than 30% PTX.16
Statistical Analysis
The study group was dichotomized based on presence or absence of PTX detected on CR. Descriptive statistics and the χ2 test were used for univariate and bivariate analysis. Sensitivity of CR for detecting PTX was calculated.
Regression Analysis
A binary logistic regression model was fit to the data. A priori determination for model inclusion was any variables with significance up to and including 0.1 on bivariate analysis. As is customary, the maximum number of variables included was capped at 1 per 10 positive cases of PTX on radiograph. A Hosmer-Lemeshow goodness of fit variable was used to determine fitness of the model with a significant result indicating overfitting.
Power Analysis
With equal size groups, this study has the power of 80% to detect a 10% absolute difference in single predictors of PTX on CR with 45 subjects in each group. Alpha was set at 0.05.
Kappa Scoring
To ensure that data collection was consistent among authors, κ statistics were calculated for a proportion of the data. We chose 20% as a representative sampling.
Human Subjects Approval
The study underwent review by our human subjects protection organization and was approved as exempt. No consent was required as no identifiers were collected.
RESULTS
Over the 2-year study period, the number of recorded trauma patients presenting to the ED was 6,732. Of these, 2,694 had some kind of chest trauma with anything coded from thorax with Abbreviated Injury Scale codes between 40009.9 to 451022.5. The search was done with a word search function. Also searched were the keywords chest tubes (34.04) and CT of the chest (87.41). A total of 367 patients had chest CTs done.
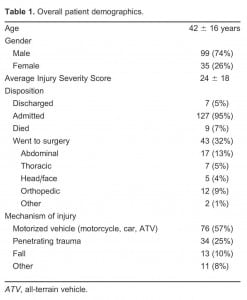
There were 134 CT-documented PTX included in the study. At our institution, CT is the gold standard for those with PTX related to trauma; therefore, standard of care at our institution is to obtain a chest CT. Mean age of patients was 42, and 74% were men. PTX was detected on CR for 66 of 134 (sensitivity = 49%). See Table 1.
The majority of the PTXs were small (52%), as illustrated in Table 2. PTX was detected on CR in 30% of small PTXs, 35% of moderate PTXs, and 33% of large PTXs. See Table 3. Chest tubes were inserted in 89% of patients with PTX detected on CR and 44% of patients with PTX detected only on CT (difference 45%; 95% confidence interval 30, 58).
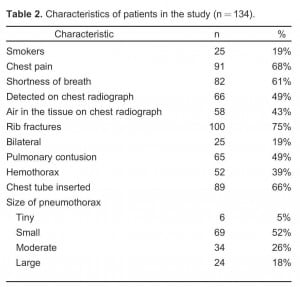
Characteristics of patients in the study (n = 134).
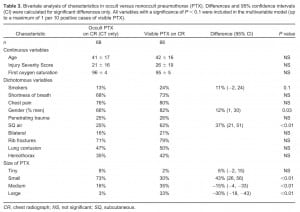
Bivariate analysis of characteristics in occult versus nonoccult pneumothorax (PTX). Differences and 95% confidence intervals (CI) were calculated for significant differences only. All variables with a significance of P < 0.1 were included in the multivariable model (up to a maximum of 1 per 10 positive cases of visible PTX).
Comparing patients with and without PTX on CR, there were no significant differences in shortness of breath (68% vs 73%, respectively) or chest pain (76% vs 80%, respectively). There were also no relationships between PTX detected on CR and age, gender, penetrating versus blunt injury, bilaterality of the PTX, presence of rib fractures, or presence of lung contusion or hemothorax on CT. We found differences up to a significance of 0.1 in bivariate analysis for smokers, male gender, presence of SQ air, and larger-sized PTXs. These variables were then entered into the multivariable analysis. See Table 3.
When adjusting for all variables on bivariate analysis that reached a significance level of 0.1, significant predictors of a PTX detected on CR were air in the tissue (adjusted odds ratio [OR] = 3.8; P = 0.02) and PTX size (adjusted OR = 51.0; P < 0.01) for a large PTX compared to a tiny PTX. See Table 4.
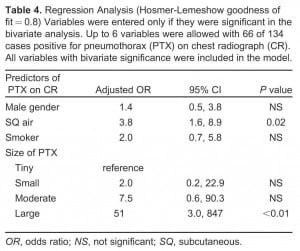
Regression Analysis (Hosmer-Lemeshow goodness of fit = 0.8) Variables were entered only if they were significant in the bivariate analysis. Up to 6 variables were allowed with 66 of 134 cases positive for pneumothorax (PTX) on chest radiograph (CR). All variables with bivariate significance were included in the model.
In approximately 20% of patients, a different author was responsible for checking data entry for agreement. No changes were made in the data based on this reexamination of the extraction techniques. The κ statistic for agreement was excellent at 0.88 (0.87 to 1).
Missing Data
For 17 (13%) subjects, information on presence of chest pain and shortness of breath was missing. For 1 (0.7%) patient, information on the size of the PTX was missing. There were no missing entries for other data.
DISCUSSION
Overall, we found that the only significant predictors of occult PTX on CR were SQ air in the tissue on CR and size of PTX. Air in the tissue was a CR finding, and PTX size was a CT finding, so neither would be available when a patient presents.
None of the variables available before the CR was performed were helpful in predicting PTX on CR. These variables of patient condition and demographics were not predictive of the presence of OPTX. Neither symptoms of chest pain nor shortness of breath predicted whether we could see a PTX on CR. We conclude that in patients with major trauma and a high Injury Severity Score (ISS), CT is necessary to ensure the diagnosis of PTX regardless of symptoms.
Even after a CR was completed, we found that it was not a sensitive indicator of PTX on CT. Air in the tissue was the only variable on the CR that could have helped predict OPTX and that only increased the odds fourfold. In a previous study the rates were similar.1 We conclude that CT is necessary to ensure PTX diagnosis regardless of CR findings.
An important finding of this study was that no PTX was apparent on half of the CRs and yet 44% of these patients with OPTX required chest tube placement. Again, this suggests that the CR was of little value in affecting the management of these patients. Other studies have shown similar rates.2,10 Other studies have shown that it is acceptable to expectantly manage such cases, unless positive pressure ventilation (PPV) is necessary.9Up to 38% of OPTXs may progress with PPV.15 This is similar to the findings of a previous study. The other side of the issue is that because the CR was positive in about half of the patients, it could have led to an earlier placement of a chest tube in all of these patients. However, Yadav et al17 found that chest tube placement in patients with OPTX was not safer or any more effective than managing with observation alone. There is nothing in the literature to suggest that an earlier placement of a chest tube in patients with OPTX leads to better outcomes.
As in most other studies of PTX most of our patients were men and most causes were secondary to blunt trauma. A large percentage of our patients had chest pain and/or shortness of breath (>60%). None of these variables, however, were associated with a higher incidence of OPTX.
Our results suggest either the need for routine CT or another approach to OPTX detection. Some studies have used different approaches to detect PTX without requiring CT, including use of oblique chest radiographs18 and use of ultrasound.12–14,19,20 Ultrasound as part of a routine FAST exam showed a high sensitivity for detecting OPTX and is safe and expedient for diagnosing them.19,20 We did not routinely use ultrasound in our ED, except in patients with hypotension.
Most of the OPTXs treated in our ED were categorized as small. There is much controversy in these measurements, however, and they can be hospital dependent.21–23 Only about a third of the PTXs in each of these categories were detected on initial CR in our study. This is a surprising finding as almost a third of large PTXs detected on CT were also detected on initial CR. Location of the PTX or patient positioning may have been a factor.
For most of the patients in our study, the initial CR was done with the patient in a supine position. Other techniques have been used to delineate PTXs, including the use of supine films, expiratory films, lordotic views, oblique views, or lateral CRs.18 Even though these methods may demonstrate PTX on initial CR, this is often not possible in the trauma patient since the trauma patient can be unstable and is, therefore, impractical. Matsumoto et al18 describe use of an oblique view of the chest which revealed a visceral pleural line, consistent with PTX. Signs on film taken with the subject in the supine position that may indicate PTX include deepening of the costophrenic angle (deep sulcus sign) or the presence of 2 diaphragm/lung interfaces (double diaphragm sign). Sensitivity is poor in detecting PTX, however.18 Other studies have shown that ultrasound can be a useful approach as it is a good tool for determining movement of the diaphragm, which is absent in many PTXs.12–14 However, this technique was not routinely used in our ED in 2008–2010.
A recent study comparing ultrasound to CR and CT showed that ultrasound was more sensitive (82%) and specific (100%) than CR in diagnosing PTX. More PTXs were detected by CT. The conclusion of the study is that ultrasound of the lungs should be included in the FAST.23
For our regression analysis, predictors of PTX included large size and air in the tissue. When corrected for these variables, gender and smoking history, which were significant in univariate analysis, fell out of the model. These demographic and social factors (ie, gender and smoking history) are not helpful in predicting a diagnosis at a patient’s initial presentation. Therefore, it is still essential to obtain an image that will be useful for diagnosing PTX, and CT and ultrasound may be the best tests for this purpose.
In summary, symptoms, patient demographics, and mechanism of injury were not useful in detecting OPTX. Only the presence of air in the tissue on CR was a valuable clue to the presence of an OPTX. Size of the PTX matters in detecting it on CR, and there is still controversy on whether the smaller PTXs should be treated or observed. However, CT use must either continue to be the gold standard or other alternatives must be found for PTX detection.
LIMITATIONS
This study was retrospective in nature, some of the patients with PTX may have been missed. It is unknown how many were missed because they only received a plain CR and did not undergo CT; however, this is potentially rare.
The study was limited because we did not evaluate any alternative methods of PTX detection. At the time of the study, the institution did not routinely do ultrasound and it is not part of our routine FAST exam. Ultrasound may prove to be a method for decreasing the number of CTs necessary in the future.
Data extraction may have been flawed because we used 2 abstractors. However, we made every effort to ensure that both abstractors were given equivalent education. In addition, we evaluated about 20% of the data for comparison and found excellent κ statistic agreement among data extractors.
No known records were left out because of incomplete data. Some may have been missed because the injury was improperly categorized; however, it is likely a very small number. It is impossible to determine numbers that were not entered correctly or were never entered.
The need for a chest tube was based on the clinical judgment of the individual clinicians. Studies have shown that clinical observation of a patient with a small PTX is possible. Because of the retrospective nature of this study, it is not possible to determine if these patients could have been observed.
At our institution ultrasound is available in the trauma area; however, lung evaluation for PTX is not typically part of the routine evaluation for patients. Ultrasound is a quick, noninvasive, and accurate method of detecting a PTX. Whether chest tubes would be placed or not based on an ultrasound diagnosis alone is uncertain.
The study was based on chart review, and cases may have been missed or study data miscoded when the chart was recorded. The study did not evaluate cases in which a PTX was found on CR and for which a CT was not obtained; however, in our institution the overwhelming practice is to proceed to CT if there is possibility of chest injury.
The ISS values in our patients varied widely from 6 (minor) to 42 (very severe). This may have skewed results considerably in our study. In addition, unidentified confounder variables likely exist that were not included in our regression analysis.
CONCLUSION
Factors associated with PTX on CR included air in the soft tissue on CR and size of the PTX. Even when PTX is not apparent on CR, 44% of these PTXs received placement of a chest tube.
Footnotes
The authors would like to acknowledge the generosity of the George Valente Foundation contributions that helped make this research possible. The foundation had no role in the design of the research.
Supervising Section Editor: Michael Burg, MD
Submission history: Submitted April 28, 2011 ; Revision received August 14, 2011 ; Accepted September 19, 2011
Full text available through open access at http://escholarship.orgfucfuciem_westjem
DOl: 1 0.5811fwestjem.2011.9.6781
Address for Correspondence: Amy A. Ernst, MD, University of New Mexico, Department of Emergency Medicine, MSC10 5560 1, Albuquerque, NM 87131
E-mail: aernst56@aol.com
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Wolfman NT, Myers WS, Glauser SJ, et al. Validity of CT classification on management of occult pneumothorax: a prospective study. AJR Am J Roentgenol. 1998;171:1317–1320.[PubMed]
2. Bridges KG, Welch G, Silver M, et al. CT detection of occult pneumothorax in multiple trauma patients. J Emerg Med. 1993;11:179–186. [PubMed]
3. Neff MA, Monk JS, Peters K, et al. Detection of occult pneumothoraces on abdominal computed tomographic scans in trauma patients. J Trauma. 2000;49:281–285. [PubMed]
4. Enderson BL, Abdalla R, Frame SB, et al. Tube thoracostomy for occult pneumothorax: a prospective randomized study of its use. J Trauma. 1993;35:726–730. [PubMed]
5. Shorr RM, Crittenden M, Indeck M, et al. Blunt thoracic trauma: analysis of 515 patients. Ann Surg. 1987;206:200–205. [PMC free article] [PubMed]
6. Deakin CD, Davies G, Wilson A. Simple thoracostomy avoids chest drain insertion in pre-hospital trauma. J Trauma. 1995;39:373–374. [PubMed]
7. Holmes JF, Brant WE, Bogren HG, et al. Prevalence and importance of pneumothoraces visualized on abdominal computed tomographic scan in children with blunt trauma. J Trauma. 2001;50:516–520. [PubMed]
8. Guerrero-Lopez F, Vasquez-Mata G, Alcazar-Romero P, et al. Evaluation of the utility of computed tomography in the initial assessment of the critical care patient with chest trauma. Crit Care Med. 2000;28:1370–1375. [PubMed]
9. Ball CG, Hameed SM, Evans D, et al. Occult pneumothorax in the mechanically ventilated trauma patient. Can J Surg. 2003;46:373–379. [PMC free article] [PubMed]
10. Brazel KJ, Stafford RE, Weigelt JA, et al. Treatment of occult pneumothoraces from blunt trauma. J Trauma. 1999;46:987–991. [PubMed]
11. Plurad D, Green D, Demetriades D, et al. The increasing use of chest tomography for trauma: is it being overutilized. J Trauma. 2007;62:631–635. [PubMed]
12. Zhang M, Liu ZH, Yang JX, et al. Rapid detection of pneumothorax by ultrasonography in patients with multiple trauma. Crit Care. 2006;10:R112–R118.[PMC free article] [PubMed]
13. Soldati G, Testa A, Sher S, et al. Occult traumatic pneumothorax: diagnostic accuracy of lung ultrasonography in the emergency department. Chest. 2008;133:204–211. [PubMed]
14. Blaivas M, Lyon M, Duggal S. A prospective comparison of supine chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad Emerg Med.2005;12:844–849. [PubMed]
15. Baumann MH, Noppen M. Pneumothorax. Respirology. 2004;9:157–164. [PubMed]
16. Collins CD, Lopez A, Mathie A, et al. Quantification of pneumothorax size on chest radiographs using interpleural distances. AJR Am J Roentgenol. 1995;165:1127–1130.[PubMed]
17. Yadav K, Jalili M, Zehtabchi S. Management of traumatic occult pneumothorax.Resuscitation. 2010;81:1063–1068. [PubMed]
18. Matsumoto S, Kishikawa M, Hayakawa K, et al. A method to detect occult pneumothorax with chest radiography. Ann Emerg Med. 2011;57:378–381. [PubMed]
19. Volpicelli G. Sonographic diagnosis of pneumothorax. Intensive Care Med.2011;37:224–232. [PubMed]
20. Volpicelli G, Cardinale L, Berchialla P, et al. A comparison of different diagnostic tests in the bedside evaluation of pleuritic pain in the ED. Am J Emerg Med. [published online ahead of print January 27, 2011] [PubMed]
21. Chan SS. Estimation of size of pneumothorax under the new BTS guidelines. Thorax.2004;59:356–357. [PMC free article] [PubMed]
22. Kelly AM, Druda D. Comparison of size classification of primary spontaneous pneumothorax by three international guidelines: a case for international consensus?Respir Med. 2008;102:1830–1832. [PubMed]
23. Nagarsheth K, Kurek S. Ultrasound detection of pneumothorax compared with chest x-ray and computed tomography scan. Am Surg. 2011;77:480–483. [PubMed]