| Author | Affiliation |
|---|---|
| Peter Hallas, MD | Copenhagen University Hospital, Department of Pediatric Anesthesia, Rigshospitalet, Denmark |
| Mikkel Brabrand, MD | Hospital of Southwest Denmark, Department of Medicine, Esbjerg, Denmark |
| Lars Folkestad, MD | Hospital of Southwest Denmark, Department of Endocrinology, Esbjerg, Demark |
Introduction
Methods
Results
Discussion
Limitations
Conclusion
Abstract
Introduction:
Intraosseous access (IO) is indicated if vascular access cannot be quickly established during resuscitation. Complication rates are estimated to be low, based on small patient series, model or cadaver studies, and case reports. However, user experience with IO use in real-life emergency situations might differ from the results in the controlled environment of model studies and small patient series. We performed a survey of IO use in real-life emergency situations to assess users’ experiences of complications.
Methods:
An online questionnaire was sent to Scandinavian emergency physicians, anesthesiologists and pediatricians.
Results:
1,802 clinical cases of IO use was reported by n=386 responders. Commonly reported complications with establishing IO access were patient discomfort/pain (7.1%), difficulties with penetration of periosteum with IO needle (10.3%), difficulties with aspiration of bone marrow (12.3%), and bended/broken needle (4.0%). When using an established IO access the reported complications were difficulties with injection fluid and drugs after IO insertion (7.4%), slow infusion (despite use of pressure bag) (8.8%), displacement after insertion (8.5%), and extravasation (3.7%). Compartment syndrome and osteomyelitis occurred in 0.6% and 0.4% of cases respectively.
Conclusion:
In users’ recollection of real-life IO use, perceived complications were more frequent than usually reported from model studies. The perceived difficulties with using IO could affect the willingness of medical staff to use IO. Therefore, user experience should be addressed both in education of how to use, and research and development of IOs.
INTRODUCTION
Intraosseous access (IO) is indicated in critically ill patients if vascular access cannot be quickly established.1 Complication rates with IO are estimated to be low, a notion that seems to come mainly from small patient series, model or cadaver studies, and case reports.2–10 By convention, these studies often define “success rate” as insertion rate, i.e. the proportion of needles that penetrate the cortex. This success rate is usually high, 71–100%.2–10
IO is a relatively rare occurrence in many settings, and healthcare staff might be inexperienced in the procedure or unfamiliar with the available IO device. Using IO in real-life situations could pose a different set of challenges (e.g., moving patient, high-stress situation, not much room to work around, many people involved in resuscitation) compared to the often controlled settings of small patient series, model or cadaver studies. Therefore, it is possible that the rate of experienced complications is much higher in real-life emergency situations than described in small patient series, model or cadaver studies.
Users’ recollection of complications with using a medical device will influence their willingness to use it in the future. Thus, information on users’ experience with complication rates in real-life use of IO could be used to identify issues for improvements in education and device design. For this purpose, we collected information on what Scandinavian emergency physicians, anesthesiologists and pediatricians experienced with real-life IO use.
METHODS
We collected cases of users’ experiences with cases of IO use by distributing a link to an online survey via e-mail to members of the Danish Society for Emergency Medicine (DASEM) (n=394 members). Responders were asked to recall all the cases where they had used IO in a clinical situation. Thus, the survey focused on the users’ recollection of IO use. It did not involve review of patient charts, and it was not a prospective registration of cases. We used the survey software LimeSurvey (freely available at http://www.limesurvey.org). Additional responses were obtained by asking other Scandinavian emergency medicine, pediatric and anesthesiology societies and interest groups to distribute a link to the questionnaire to their members. These were the Danish Society for Anaesthesia and Critical Care, the Danish Paediatric Society, the Norwegian Society for Anaesthesiology, the Norwegian Society of Paediatricians, clinical staff at the Norwegian Air Ambulance, Norwegian Association on Trauma, Emergency and Disaster Medicine, the Swedish Society for Emergency Medicine, and the Swedish Society for Anaesthesia and Intensive Care.
The survey was designed to collect detailed information on all clinical cases where the responders personally had used IO. We included responders in this analysis if they had real-life experience with IO (i.e., they had themselves placed or tried to place an IO). The responders were given detailed questions on successful and unsuccessful IO attempts, type of IO device used in each case, and complications or technical difficulties with the specific device. The questionnaire also included sections on training in IO and barriers to IO use in clinical settings; results on these aspects are published elsewhere.11,12 We designed the survey so that no single patient could be identified from the data entered. Therefore, data on age groups, anatomical sites used and indication for IO placement were not available for all cases. The questionnaire had been tested by 5 doctors working in emergency care prior to distribution. None of them was involved in the research group. We used their feedback to improve the design of the survey.
The survey did not fall under jurisdiction of the Danish Act on Processing of Personal Data and – as it was not a clinical trial – approval of an ethics committee was not relevant.
We performed statistical analysis with Chi-test for trend and Fisher’s Exact Test.
RESULTS
We received 761 responses to the questionnaire; n=388 responders met the inclusion criteria. We excluded 2 of these due to gross inconsistencies in the answers. The response rate for DASEM members was 37.3%.
Perceptions of IO use in Scandinavia
The 386 responders reported a total of 1,802 clinical cases of IO use. Information on age of the patient was available for 1,719 cases. Of these, 774 (45.0%) were pediatric patients (under age 15) and 380 (49.1% of pediatric patients) were less than 2 years old. The ratio of needle use per patient was 1.17 for adults (15 years or older), 1.19 for children between 6 to 14 years (both inclusive), 1.18 for children between 2 and 5 years (both inclusive), and 1.14 for children under 2 years. (Chi square for trend did not detect a significant difference: p=0.98.)
The indications for IO use were as follows: Cardiac arrest (28.4%; n=486), hemorrhage (23.4%; n=407), dehydration (16.7%, n=286), sepsis (13.4%, n=230), convulsions (2.7%; n= 46), poisoning (2.2%, n=37), and other clinical situations (12.8%, n=219). Information on indication for IO use was available for 1,711 cases (95.0%).
IO had been placed in the tibia in 1,420 cases (89.7%), in the humerus in 54 cases (3.4%), in the iliac crest in 34 cases (2.1%), in the sternum in 27 cases (1.7%), in the femur in 20 cases (1.3%), and other sites in 28 cases (1.8%). Information on anatomical site was available for 1583 cases (87.8%).
The IO-devices used were the following: EZ-IO in 861 cases (47.8%), Cook-Surfast in 418 cases (23.2%), B.I.G. in 255 cases (14.2%), and unspecified equipment in 268 cases (14.9%).
Users’ recollection of complications
Table shows the overall frequency of reported complications and complication rates listed by device. In addition to Table, users reported “Miscellaneous complications” in n=36 cases (not specified).
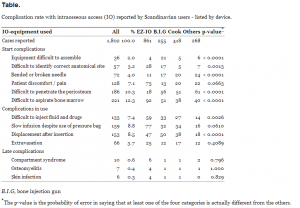
Complication rate with intraosseous access (IO) reported by Scandinavian users – listed by device.
DISCUSSION
In this study, IO users reported complications with real-life IO use from a multitude of settings, including prehospital, emergency departments (ED) and intensive care units. The rate of many of these complications will not be revealed in the controlled environment in simulation and cadaver studies for example, because they lack the difficulties with assembling equipment in extremely stressful situations and cannot be assessed for infection rates. Therefore, reports from a large number of cases of IO use are useful to get an estimate of complication rates in real-life situations (with the limitations of the study in mind, discussed below). More importantly, the users’ recollection of difficulties with IO use could affect the willingness to use IO in future cases – even if the users don’t recall every detail of the previous cases.
Healthcare staff reported a wide range of difficulties with the use of IO. These difficulties might render the IO useless in an emergency situation. For example, the penetration through the cortex might have been successful, but the IO itself could be useless because of immediate dislocation. Thus, IO might not always be experienced as a straightforward procedure by healthcare staff when it is performed in real-life emergency situations.
The study supports 2 common notions about IO. Firstly, that the rate of serious, late complications (i.e. compartment syndrome and ostemyolitis) is low. Indeed, the rate of 0.4–0.6% is remarkably close to the rates found in the 2 previous large-scale studies using this as a primary end-point. Secondly, that needle insertion rates per se can be tranferred from IO model or simulation studies to real-life situations: In about 90% of the reported cases in this study only 1 needle was used which corresponds to the needle insertions success rate often reported from model and cadaver studies
The findings have implications for future education, design and research in IO usage. Doctors might be reluctant to use IO again if they remember earlier cases of IO use as difficult or as resulting in unexpected complications. Therefore, the difficulties reported by IO users should be considered when training healthcare staff in IO. It is the impression of the authors that many IO courses focus on the insertion step of the procedure. But perhaps educators to a wider extent should prepare healthcare staff for the most common difficulties in the later parts of the procedure as well and focus on measures to prevent and deal with these issues. Furthermore, as can be seen from the table, specific types of IO device designs seem to be prone to certain types of complications. Optimally, designers of IO devices should learn from these differences. In addition, the relatively high rate of difficulties after insertion should be kept in mind when doing research in IO, including simulation studies. Measuring IO success rate merely as “penetration through cortex” could miss clinically relevant complications.
LIMITATIONS
The response rate for this questionnaire was 37% for DASEM members. This was estimated by comparing e-mail addresses from the survey with the membership register of DASEM. Some responders did not wish to state their e-mail address, so the response rate is a minimum estimate. For the rest of the societies a response rate could not be reliably estimated for reasons discussed elsewhere.11 A low response rate could give a risk of selection bias, but a response rate of around 40% is actually considered average for e-mail surveys.15
Because this study was founded on users’ recollection of events rather than a review of patient databases or charts there was an obvious risk of recall bias. Thus, the complication rates reported by the users are not necessarily identical to the complication rates that would have been found in a prospective study or even in a retrospective study of patient charts. This was especially true for late complications, e.g., infections, since a doctor placing the IO in the prehospital setting or the ED often will have limited information on long-term complications. However, the data still accurately represent how users recall the cases.
CONCLUSION
In users’ recollection of real-life IO use, the overall rate of complications was higher than usually reported from model and cadaver studies. The relatively high rate of difficulties after insertion should be addressed when the procedure is taught. Research on IO devices should focus on all stages of IO use. Focus only on “penetration through cortex” as a measure of IO success rate could miss clinically relevant complications.
Footnotes
Address for Correspondence: Peter Hallas, Department of Pediatric Anesthesia, Copenhagen University Hospital, Rigshospitalet, Blegdamsvej 3, 2100 Ø, Denmark. Email: hallas@rocketmail.com.
Submission history: Revision received February 19, 2012; Submitted December 11, 2012; Accepted January 29, 2013
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1 Luck RP, Haines C, Mull CC Intraosseous access. J Emerg Med. 2010; 39:468-475
2 Ngo AS, Oh JJ, Chen Y Intraosseous vascular access in adults using the EZ-IO in an emergency department. Int J Emerg Med. 2009; 11:155-160
3 Gerritse BM, Scheffer GJ, Draaisma JM Prehospital intraosseus access with the bone injection gun by a helicopter-transported emergency medical team. J Trauma. 2009; 66:1739-1741
4 Fiorito BA, Mirza F, Doran TM Intraosseous access in the setting of pediatric critical care transport. Pediatr Crit Care Med. 2005; 6:50-53
5 Jun H, Haruyama AZ, Chang KS Comparison of a new screw-tipped intraosseous needle versus a standard bone marrow aspiration needle for infusion. Am J Emerg Med. 2000; 18:135-139
6 Anderson TE, Arthur K, Kleinman M Intraosseous infusion: success of a standardized regional training program for prehospital advanced life support providers. Ann Emerg Med. 1994; 23:52-55
7 Frascone RJ, Jensen J, Wewerka SS Use of the pediatric EZ-IO needle by emergency medical services providers. Pediatr Emerg Care. 2009; 25:329-332
8 Calkins MD, Fitzgerald G, Bentley TB Intraosseous infusion devices: a comparison for potential use in special operations. J Trauma. 2000; 48:1068-1074
9 Levitan RM, Bortle CD, Snyder TA Use of a battery-operated needle driver for intraosseous access by novice users: skill acquisition with cadavers. Ann Emerg Med. 2009; 54:692-694
10 Waisman M, Waisman D Bone marrow infusion in adults. J Trauma. 1997; 42:288-293
11 Hallas P, Brabrand M, Folkestad L Reasons for not using intraosseous access in critical illness. Emerg Med J. 2010;
12 Hallas P, Folkestad L, Brabrand M How many training modalities are needed to obtain procedural confidence in intraosseous access? A questionnaire study. Eur J Emerg Med. 2011; 6:360-362
13 Heinild S, Søndergaard T, Tudvad F Bone marrow infusion in childhood: Experiences from a thousand infusions. J Pediatr. 1947; 30:400-412
14 Rosetti VA, Thompson BM, Miller J Intraosseous infusion: an alternative route of pediatric intravascular access. Ann Emerg Med. 1985; 14:885-888
15 Sheehan KB E-mail Survey Response Rates: A Review. Journal of Computer-Mediated Communication. 2001; 6


