| Author | Affiliation |
|---|---|
| Sarah T. Selby, DO | Albert Einstein College of Medicine, Jacobi Medical Center, Department of Emergency Medicine, Bronx, New York |
| Marianne Haughey, MD | Albert Einstein College of Medicine, Jacobi Medical Center, Department of Emergency Medicine, Bronx, New York |
Case report
Discussion
Conclusion
ABSTRACT
Dysfunctional uterine bleeding (DUB) is a common presentation in the emergency department and has a wide differential. Most presentations of DUB are in hemodynamically stable patients and can be evaluated as an outpatient. Uterine arteriovenous malformation (AVM) is one presentation that can result in a life-threatening medical emergency with unexpected sudden and massive vaginal bleeding. We describe a case of a 24-year-old female with sudden heavy vaginal bleeding requiring a blood transfusion, ultrasound evidence of uterine AVM, and a treatment method of expectant management using an intrauterine device in an attempt to preserve fertility.
CASE REPORT
A 24-year-old female, G3P0030 with a history of depression presented to the emergency department (ED) for evaluation of abrupt heavy vaginal bleeding. The patient noticed a sudden gush of blood between her thighs, prompting her visit to the ED. She had 2 days of light spotting preceding this sudden heavy bleeding, as well as irregular menses ever since a missed abortion 4 months prior. She had 2 elective terminations of pregnancies in the preceding year secondary to severe hyperemesis gravidarum. During this episode of sudden heavy vaginal bleeding, she also complained of mild suprapubic cramping, but no associated nausea, vomiting, dizziness, chest pain, or shortness of breath.
Upon presentation to the ED, approximately 30 minutes after the bleeding began, the patient’s triage vital signs showed a blood pressure of 123/81 mmHg, pulse of 102, respiratory rate of 20, pulse oximetry of 100% on room air, oral temperature of 98.7°F, and a negative rapid urine pregnancy. Intravenous access was obtained and blood was drawn for laboratory analysis. Upon evaluation, she was awake, alert and oriented ×3, and she was neither pale nor diaphoretic. She was bleeding through her sanitary napkins onto her clothes, but walked with ease to the exam table without any shortness of breath. Her abdomen had normoactive bowel sounds and was soft and non-tender. She had normal external genitalia with no signs of trauma. Her vaginal exam revealed copious dark blood and clots, with no trauma noted. There was approximately 300 mL of blood on the exam room floor. Her cervix had no lesions and clots were noted in the os. On bimanual exam, she had a closed cervical os, no cervical motion tenderness, and no adnexal tenderness.
Her laboratory studies, including a complete blood count (CBC), electrolyte panel, coagulation studies, and thyroid function tests ,were unremarkable. She had a hemoglobin (Hgb) of 12.1 g/dL and hematocrit (Hct) of 37.1%. A point-of-care ED transvaginal ultrasound showed an enlarged endometrial cavity containing irregularities with an area of pulsatile flow via Doppler, no free fluid, and normal ovaries. Despite receiving an initial 2-liter bolus of intravenous crystalloid, the patient developed symptoms of postural hypotension with average blood pressures at approximately 100/60 mmHg. Her heart rate remained in the nineties; however, she continued to have persistent heavy vaginal bleeding. The decision was made to admit the patient to the gynecology service for possible surgical intervention of massive dysfunctional uterine bleeding, and further evaluation of an enlarged endometrial cavity.
During her admission, and 9 hours after presentation, a repeat CBC due to heavy vaginal bleeding showed a 3-point drop in Hgb to 8.8 g/dL and Hct to 26.6%. She was transfused 4 units of packed red blood cells. On hospital day 2, her formal sonogram showed an enlarged endometrial cavity measuring 26 mm transversely with multiple hypertrophied vessels within the myometrium at the endometrial junction showing low resistance arterial flow, but no active flow within the endometrial cavity, suggesting a diagnosis of uterine arteriovenous malformation (AVM) (Figures 1 and 2). A magnetic resonance imaging angiogram (MRI/A) of the pelvis showed an amorphous collection of vessels arising from the pelvic arterial branches confirming the ultrasound diagnosis of multiple parametrial AVM or pseudoaneurysms (Figure 3).
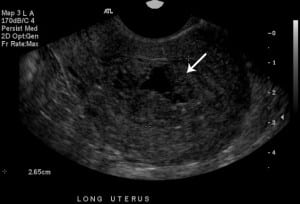
Ultrasonography of uterus in long axis showing enlarged endometrial cavity (2.65 cm) with irregular clot.
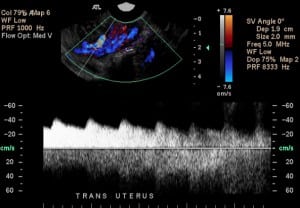
Ultrasonography of uterus in transverse axis with color doppler flow showing pulsatile flow inside the endometrial cavity.
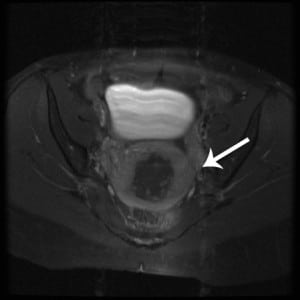
Magnetic resonance imaging of uterus in transverse axis demonstrating amorphous collection of vessels with fluid in the endometrial cavity.
On hospital day 3, she had minimal vaginal bleeding, normal vitals, and stable blood counts (Hgb 9 g/dL and Hct 27%). After a multidisiciplinary discussion involving gynecology and interventional radiology, the patient declined surgical intervention, including uterine embolization of the AVM, and preferred to try medical management in an attempt to preserve her fertility. A hormonal intrauterine device (IUD) was placed and the patient was discharged with strict instructions on when to return to the hospital if the bleeding returned.
DISCUSSION
Uterine AVMs are a rare cause of uterine bleeding with fewer than 100 cases reported.1 The true incidence is unknown, but with increased use of ultrasound to evaluate abnormal vaginal bleeding, O’Brien et al2 propose a rough predicted incidence of 4.5%. AVMs consist of an abnormal growth and connection between arteries and veins without a capillary bed, creating areas of high and low flow, which are fragile and prone to bleeding. 3 Uterine AVMs most commonly present with sudden heavy vaginal bleeding and can be congenital or acquired. Congenital AVMs form through disturbances in angiogenic development creating multiple connections between arteries and veins that tend to be deeper in surrounding tissue. Acquired AVMs form smaller arteriovenous fistulas that occur as a complication of uterine surgery or curettage, gestational trophoblast disease, choriocarcinoma, and infection.4
In a patient with massive vaginal bleeding, especially in the presence of hemodynamic instability, it is important to initiate aggressive resuscitation with intravenous fluids and early use of blood products.5 Temporizing measures, such as intrauterine tamponade with a foley catheter, can be performed in the ED to treat life-threatening vaginal hemorrhage.6 In the unstable patient, the treatment regimens include dilation and curretage (D&C), intravenous estrogen, uterine artery embolization, and hysterectomy.7 Uterine AVMs frequently cause sudden massive bleeding, and in an unstable patient appropriate diagnosis is important because emergent treatment with D&C can worsen the underlying condition, leading to profuse uterine hemorrhage, shock, and potentially hysterectomy.2
Traditionally, uterine AVMs were diagnosed after hysterectomy with histopathologic evidence of the arteriovenous fistulas.3 In the present day, angiography has become the gold standard for diagnosis of uterine AVMs with the added benefit of the ability to deliver treatment through embolization.8 Ultrasonography, computed tomography (CT) and MRI are being used more frequently as initial diagnostic modalities.8 Ultrasonography is becoming the preferred method for diagnosing AVMs, reserving angiography for planned therapeutic embolization or prior to surgical intervention.
As ultrasonography is becoming more readily available in the ED, bedside ultrasonography by the emergency physician or consulting gynecologist can aid in the initial diagnosis of uterine AVMs. Gray scale ultrasonography will often show nonspecific heterogenous or anechoic tortuous spaces in the myometrium.9 Color and spectral Doppler ultrasonography shows further detailing of a tangle of vessels producing a “color mosaic” pattern with multidirectional high and low velocity flow.2,10 Ultrasonography has also been used to demonstrate the efficacy of treatment by evaluating for resolution of AVMs within 24 hours of embolization.2
Treatment of uterine AVMs remains controversial, often with great concern for fertility. Hysterectomy remains the definitive treatment, especially in a symptomatic patient without desired fertility.4 A minimally invasive approach through angiographic embolization of the AVM, which has potential to preserve fertility, is currently the preferred treatment. In 1997, 10 years after the first successful uterine AVM embolization, there were only 5 pregnancies documented.4 A review of current literature to 2004 shows slightly more successful pregnancies following embolization at a mere 10 pregnancies; however, this still does not demonstrate adequate fertility preservation.8 Congenital AVMs tend to be treated with hysterectomy or embolization as they pose a higher risk of recurrent menorrhagia. And finally, in the correct clinical setting, conservative management is another treatment modality, typically offered to cases of acquired AVMs. Patients with one episode of bleeding and hemodynamic stability can be offered treatment with combined oral contraceptive pills (OCP).11 Timmerman et al12 report 8 cases of spontaneous resolution of acquired AVMs following OCP use or expectant management, improving the chances of preserved fertility. Even more case reports in the current literature show resolution of AVMs and successful pregnancies after conservative management.13
In this case report, a fertile female presented with sudden heavy vaginal bleeding in the setting of 2 recent D&Cs. The abrupt nature and volume of bleeding indicated the need to closely monitor for signs of hemorrhagic shock, and further evaluate for acquired uterine AVMs. The point-of-care ED ultrasound demonstrated a pulsatile mass in the endometrial cavity, prompting further workup for potentially life-threatening bleeding from a uterine AVM. Once hemodynamically stable, the patient opted for conservative medical management with an IUD to provide the highest probability of preserved fertility. IUDs have not been shown to reduce the size or risk of rupture of uterine AVMs, and there are no data to suggest that insertion is contraindicated. The patient was advised that her uterine AVM was likely acquired from her prior D&Cs and they could regress spontaneously or rupture again, possibly complicating future pregnancy. Her future might involve AVM embolization or hysterectomy as a last resort.
CONCLUSION
Dysfunctional uterine bleeding is a common complaint in the ED, and in the right clinical setting, can be life threatening. When faced with a patient with sudden and massive vaginal bleeding and a history of prior uterine instrumentation, the diagnosis of uterine arteriovenous malformation should be considered. Color or spectral Doppler ultrasonography should be used to confirm the diagnosis and provide the most accurate information to the consulting gynecologist.
Footnotes
Address for Correspondence: Sarah Tolford Selby, DO, Jacobi Medical Center, Department of Emergency Medicine, 1400 Pelham Parkway South, Bronx, NY 10461. Email: sarahtselby@gmail.com.
Submission history: Revision received July 24, 2012; Submitted November 16, 2012; Accepted December 12, 2012
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Hickey M, Fraser IS. Clinical implications of disturbances of uterine vascular morphology and function. Ballieres Clin Obstet Gynaecol. 2000;14:937–951. [PubMed]
2. O’Brien P, Neyastani A, Buckley A, et al. Uterine artery malformations from diagnosis to treatment. J Ultrasound Med. 2006;25:1387–1392. [PubMed]
3. Fleming H, Ostor AG, Pickel H, et al. Arteriovenous malformations of the Uterus. Obstet Gynecol.1989;73:209–213. [PubMed]
4. Hoffman MK, Meilstrup JW, Shackelford DP, et al. Arteriovenous malformations of the uterus: an uncommon cause of vaginal bleeding. Obstetrical and Gynecological Survey. 1997;52(12):736–40.[PubMed]
5. Manning JE. Fluid and blood resuscitation. In: Tintinalli JE, Kelen GD, Stapczynski JS, editors.Emergency Medicine: A Comprehensive Study Guide. 6th ed. New York, NY: McGraw-Hill; 2004. pp. 225–31.
6. Goldrath MH. Uterine tamponade for the control of acute uterine bleeding. Am J Obstet Gynecol.1983;147(8):869–72. [PubMed]
7. Chuong CJ, Brenner PF. Management of abnormal uterine bleeding. Am J Obstet Gynecol.1996;175:787–92. [PubMed]
8. Grivell M, Reid M, Mellor A. Uterine arteriovenous malformations: a review of current literature.Obstet Gynecol Surg. 2005;60(11):761–767. [PubMed]
9. Huang MW, Muradali D, Thurston WA, et al. Uterine arteriovenous malformations: gray scale and Doppler ultrasound features with MR imaging correlation. Radiology. 1998;21:115–123. [PubMed]
10. Polat P, Suma S, Kantarcy M, et al. Color Doppler US in the evaluation of uterine vascular abnormalities. Radio Graphics. 2002;22:47–53. [PubMed]
11. Khatree MHD, Titiz H. Medical treatment of a uterine arteriovenous malformation. Obstet Gynecol.1999;39:378–380. [PubMed]
12. Timmerman D, Van den Bosch T, Peeraer K, et al. Vascular malformations in the uterus: ultrasonographic diagnosis and conservative management. Eur J Obstet Gynecol Reprod Biol.2000;92:171–178. [PubMed]
13. Degani S, Leibovitz Z, Shapiro I, et al. Expectant management of pregnancy-related high-velocity uterine arteriovenous shunt diagnosed after abortion. Int J Gynecol Obstet. 2009;106:46–49. [PubMed]


