| Author | Affiliation |
|---|---|
| Getaw Worku Hassen, MD, PhD | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
| Hossein Kalantari, MD | New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, New York, New York |
ABSTRACT
Subdural hematoma (SDH) is a rare, but life-threatening complication of spinal anesthesia. Subdural hematoma resulting from this procedure could present with vague symptoms such as chronic headache and could easily be missed. Chronic headache is one of the symptoms of chronic SDH in postpartum women. Diplopia as the presenting complaint in SDH secondary to peripartum spinal anesthesia has not, to our knowledge, been previously reported. Here, we report a case of diplopia secondary to postpartum subacute bilateral SDHs with transtentorial herniation after spinal anesthesia in a healthy primagravid 25-year-old woman. SDH can expand gradually and the initial symptoms might be subtle as in our case, despite critically high intracranial pressure.
INTRODUCTION
Spinal anesthesia has become very popular for obstetric procedures. Such use was first described in 1901.1 It involves the injection of anesthetic solution into the spinal peridural or subarachnoid space. Spinal anesthesia offers the advantage of avoiding general anesthesia and the patients’ remaining awake during the procedure.2 Complications of spinal anesthesia include hypotension, postdural puncture headache, meningitis, spinal hematoma and reversible sensory loss, and paraplegia. Subdural hematoma (SDH) and abducens palsy are uncommon complications of spinal anesthesia.3–8 Increased intracranial pressure (ICP) may manifest as chronic headache and focal neurologic deficits.9–11
CASE DESCRIPTION
A 25-year-old woman (G1, P1, A0) presented to the emergency department with a chief complaint of binocular double vision. She stated that she had delivered her first baby vaginally about 4 weeks before presentation, under spinal anesthesia. The entire pregnancy and peripartum period were uneventful except for moderate neck pain with stiffness and spasm starting shortly after delivery, for which she used ibuprofen every 6 hours for 2 to 3 days. The patient reported no headache, nausea, vomiting, change in vision, difficulty speaking, numbness, or weakness during that time. Two weeks after delivery she started noticing double vision, which progressively worsened to the point where she was having difficulty driving her car.
The review of systems was negative for fever, chills, chest pain, dyspnea, nausea, vomiting, abdominal pain, or urinary symptoms. She reported no recent or remote head trauma as well as no bleeding tendency or family history of bleeding disorders. She had not taken any medications except ibuprofen for her neck pain.
The physical examination did not demonstrate any abnormalities except horizontal diplopia on forward gaze and left lateral gaze. Diplopia disappeared on gaze to the right and when covering either eye. Visual acuity was normal when each eye was examined individually. Speech, visual field, motor, sensory, deep tendon reflexes as well as gait and coordination examinations were all unremarkable.
Laboratory tests including complete blood count, complete metabolic panel, and coagulation parameters were within normal limits (platelet count, 247 × 109/L; international normalized ratio, 1.03). Chest radiograph and electrocardiogram were unremarkable. A noncontrast computed tomography (CT) of the brain revealed large bilateral subacute SDH involving the bilateral fronto-temporo-parietal regions with compression of the lateral ventricles and transtentorial herniation (Figure, parts A through C). A CT angiography of the head and neck did not reveal any vascular abnormality.
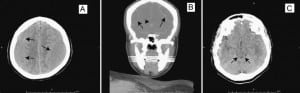
Neurology and neurosurgery services were consulted and the neurosurgical team admitted the patient for surgical intervention and further management. The patient underwent craniotomy with evacuation of bilateral SDH. No other abnormalities were noted intraoperatively, nor did any postoperative complications occur. The patient did well after the procedure with marked improvement of her symptoms. During the course of her hospital stay, the diplopia resolved completely and the patient remained asymptomatic without any neurologic deficit.
DISCUSSION
Multiple complications can occur after lumbar puncture (LP), including cerebral and spinal herniation, postlumbar puncture headache (PLPH), cranial neuropathies, nerve root irritation, low back pain, infection, and bleeding.4,9,12,13
PLPH is a frequent complication of lumbar puncture and spinal anesthesia. The mechanism that is proposed for this phenomenon is the persistent leakage of cerebrospinal fluid through the dural puncture site, leading to the caudal displacement of the brain with traction on pain-sensitive structures such as blood vessels.4,14,15 Similar mechanisms may apply traction force on the bridging veins, possibly causing a slow and constant blood leakage from these veins. The blood can accumulate over time causing an SDH and leading to symptoms such as headache or focal neurologic deficit.3,10,11,16
Contributing factors to the development of SDH include trauma, cerebral atrophy, the use of an anticoagulant after surgery, and bleeding disorders. Contributing factors to the development of PLPH are cerebral atrophy and dehydration.17,18
There is no uniform definition of PLPH, but a widely accepted definition is as follows: a constant headache appearing or worsening significantly upon assuming the upright position and resolving or improving significantly upon lying down. PLPH may persist up to 2 weeks and generally responds well to pain medications; however, occasionally, placement of epidural blood patch may become necessary. Headache from SDH may persist longer and frequently does not respond to analgesics. This headache is more likely due to increased ICP secondary to the development of subdural hygroma and subsequent SDH.19,20
Our patient did not develop the characteristic PLPH. She had no other predisposing factors for SDH, such as trauma or coagulation disorders. She only reported moderate neck pain with stiffness and spasm. She did not have any focal neurologic deficit except double vision. The double vision improved initially, but subsequently caused significant lifestyle restrictions leading to her decision to seek medical care.
Abducens palsy has been reported after LP and spinal anesthesia,13,21 but there was no peripartum or SDH association. Earlier reports of cranial nerve (CN) palsy suggested that the downward traction of the brain and compression of the CN leads to the neurologic deficit.9,21 In our patient, the downward displacement of the brain secondary to bilateral SDH may have caused compression of the cranial nerve that led to her symptom. Abducens palsy is the most common CN affected in post-LP palsy. The abducens nerve may be more sensitive to this effect, in comparison to other cranial nerves, owing to its long intracranial course. Abducens palsy usually occurs 4 to 14 days post LP and spinal anesthesia. It can be unilateral or bilateral and is usually associated with PLPH.4,9
CONCLUSION
Patients with SDH post spinal anesthesia may present with persistent headache, weakness or numbness in their extremities, or isolated cranial nerve palsy. Since a subtle focal neurologic deficit may be the initial presenting sign or symptom of a potentially devastating ICP, with or without impending herniation, physicians should maintain a high index of suspicion in such cases.
Footnotes
We thank Ms Perry and Drs Shaw and Zehtabchi for reviewing the manuscript.
Supervising Section Editor: Rick A. McPheeters, DO
Submission history: Submitted August 3, 2011; Accepted August 4, 2011
Reprints available through open access at http://escholarship.org/uc/uciem_westjem
DOI: 10.5811/westjem.2011.8.6872
Address for Correspondence: Getaw worku Hassen, MD
New York Medical College, Metropolitan Hospital Center, Department of Emergency Medicine, 1901 First Ave, New York, NY 10028
E-mail: getawh@yahoo.com
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding, sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Schneider MC, Holzgreve W. 100 Years ago: Oskar Kreis, a pioneer in spinal obstetric analgesia at the University Women’s Clinic of Basel [in German] Anaesthesist. 2001;50:525–528. [PubMed]
2. Ghirardini G, Baraldi R, Bertellini C, et al. Advantages of spinal anesthesia in abdominal gynecologic surgery. Clin Exp Obstet Gynecol. 1998;25:105–106. [PubMed]
3. Acharya R, Chhabra SS, Ratra M, et al. Cranial subdural haematoma after spinal anaesthesia. Br J Anaesth. 2001;86:893–895. [PubMed]
4. Evans RW. Complications of lumbar puncture. Neurol Clin. 1998;16:83–105. [PubMed]
5. Auroy Y, Benhamou D, Bargues L, et al. Major complications of regional anesthesia in France: The SOS Regional Anesthesia Hotline Service. Anesthesiology. 2002;97:1274–1280. [PubMed]
6. Eng RH, Seligman SJ. Lumbar puncture-induced meningitis. JAMA. 1981;245:1456–1459.[PubMed]
7. Horlocker TT. Complications of spinal and epidural anesthesia. Anesthesiol Clin North America.2000;18:461–485. [PubMed]
8. Woods WW, Franklin RG. Progressive adhesive arachnoiditis following spinal anesthesia. Calif Med. 1951;75:196–198. [PMC free article] [PubMed]
9. Liegl O. Neuroophthalmological complications through liquor leakage after surgical operation on the spinal canal for diagnostic i.e. therapeutic purposes [article in German, author’s translation] Klin Monbl Augenheilkd. 1977;171:526–530. [PubMed]
10. Slowinski J, Szydlik W, Sanetra A, et al. Bilateral chronic subdural hematomas with neurologic symptoms complicating spinal anesthesia. Reg Anesth Pain Med. 2003;28:347–350. [PubMed]
11. Woitzik J, Barth M, Tuettenberg J. Persistent postpartum headache from a chronic subdural hematoma after peridural anesthesia. Obstet Gynecol. 2006;108((3, pt 2)):808–809. [PubMed]
12. Kayacan N, Arici G, Karsli B, et al. Acute subdural haematoma after accidental dural puncture during epidural anaesthesia. Int J Obstet Anesth. 2004;13:47–49. [PubMed]
13. Kose KC, Cebesoy O, Karadeniz E, et al. Eye problem following foot surgery—abducens palsy as a complication of spinal anesthesia. MedGenMed. 2005;7:15. [PMC free article] [PubMed]
14. Pannullo SC, Reich JB, Krol G, et al. MRI changes in intracranial hypotension. Neurology.1993;43:919–926. [PubMed]
15. Strupp M, Brandt T, Muller A. Incidence of post-lumbar puncture syndrome reduced by reinserting the stylet: a randomized prospective study of 600 patients. J Neurol. 1998;245:589–592.[PubMed]
16. Baldwin LN, Galizia EJ. Bilateral subdural haematomas: a rare diagnostic dilemma following spinal anaesthesia. Anaesth Intensive Care. 1993;21:120–121. [PubMed]
17. El-Kadi H, Miele VJ, Kaufman HH. Prognosis of chronic subdural hematomas. Neurosurg Clin N Am. 2000;11:553–567. [PubMed]
18. Jonsson LO, Einarsson P, Olsson GL. Subdural haematoma and spinal anaesthesia: a case report and an incidence study. Anaesthesia. 1983;38:144–146. [PubMed]
19. Lee KS. The pathogenesis and clinical significance of traumatic subdural hygroma. Brain Inj.1998;12:595–603. [PubMed]
20. Lee KS, Bae WK, Doh JW, et al. Origin of chronic subdural haematoma and relation to traumatic subdural lesions. Brain Inj. 1998;12:901–910. [PubMed]
21. Niedermuller U, Trinka E, Bauer G. Abducens palsy after lumbar puncture. Clin Neurol Neurosurg. 2002;104:61–63. [PubMed]


