| Author | Affiliation |
|---|---|
| J. Christian Fox, MD | University of California, Irvine, Department of Emergency Medicine, Orange, California |
| Andrew G Richardson, BS | University of California, Irvine, Department of Emergency Medicine, Orange, California |
| Sarah Lopez, MD, MBA | University of Southern California, Department of Emergency Medicine, Los Angeles, California |
| Matthew Solley, MD | Poudre Valley Hospital, Department of Emergency Medicine, Fort Collins, Colorado |
| Shahram Lotfipour, MD, MPH | University of California, Irvine, Department of Emergency Medicine, Orange, California |
ABSTRACT
Introduction:
Incidental findings during ultrasound examinations occur frequently with live models in training sessions. Because of the broad scope of training sessions available, the ethics and guidelines of dealing with incidental findings in live models need to be discussed.
Methods:
We provide a case of an endovaginal ultrasound that had significant unexpected findings.
Results:
This report demonstrates an important finding uncovered during an endovaginal modeling session.
Conclusion:
Models should be notified beforehand of the possibility of an incidental finding, informed about it, made aware of potential associated costs, referred to another physician for follow-up, and provided a copy of the scans. A secure copy of the ultrasound scan should be stored for future reference.
INTRODUCTION
Incidental findings are always a possibility during any patient examination. During an endovaginal ultrasound training session, a potentially life-threatening1 malformation was discovered in a model, prompting further medical care. Similar cases will occur as ultrasonography plays an increasing role in emergency care, which will necessitate the wider use of live models for training. We will discuss the ethics and guidelines dealing with incidental findings, specifically for live models.
METHODS
A 33-year-old gravida 4, para 0, patient with no prior history of uterine abnormalities participated as a female model for an introductory bedside emergency ultrasound course. Inclusive in the course was a hands-on component for pelvic ultrasonography, whereby participants performed both transabdominal and endovaginal ultrasonography. On initial evaluation, multiple vascular structures were noted within the uterus that were unknown to the ultrasound model. In Figure 1, an endovaginal ultrasound image in the sagittal plane demonstrates a normal endometrial stripe; however, an anechoic (black) area within the uterus is identified. A subsequent color Doppler ultrasound image (Figure 2), also in the sagittal plane, demonstrates disorganized blood flow in the anechoic structure visible in Figure 1. At the completion of the course, copies of the images were given to the ultrasound model with recommendation to follow-up with her gynecologist.
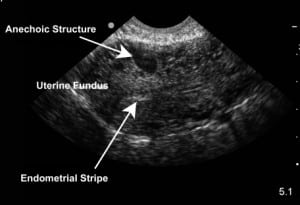
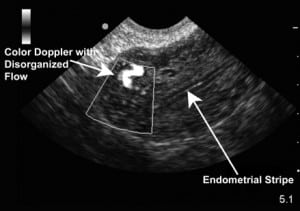
The patient’s gynecologic history is significant for 4 therapeutic abortions by dilation and curettage at ages 18, 19, 26, and 27. She began menstruating at age 12 and has very irregular and heavy cycles lasting 7 to 9 days. However, the patient did note some shortness of breath in the past 2 years, abdominal pain, or intermittent bleeding between periods.
RESULTS
The patient followed up with her gynecologist. A magnetic resonance angiogram (MRA) showed an extensive high-flow malformation involving the body and cervix of the uterus. The imaging allowed visualization of the arteriovenous malformation’s (AVM’s) blood supply, from both bilateral ovarian arteries and both bilateral uterine arteries.
The patient subsequently had left uterine percutaneous transcatheter artery embolization. Follow-up MRA demonstrated the successful occlusion of the left-sided arterial supply to the uterus but additional embolization of the AVM’s blood supply was required. Three months later the patient had a right ovarian artery embolization. The procedures were well tolerated and the patient retained function of the right ovary.
DISCUSSION
Incidental findings are bound to occur with the use of live models for ultrasound training, and emergency physicians (EP) would benefit from guidelines to assist them in dealing with such findings. There is little to no literature concerning findings in models, but Mills et al2 showed that 9.1% of patients who underwent abdominal ultrasonography also had an incidental finding. Other literature reveals numerous incidental findings in patients, including intrauterine pregnancies, gallstones, various cysts, carcinomas, and arteriovenous variations/malformations, to name a few. EPs need to be prepared to deal with incidental findings in models, as live models have been used frequently in the past decade in scientific meetings.3 The prevalence of incidental findings, combined with frequent use of models in training, adds to the importance of this issue.
The questions raised concerning incidental findings are diverse. They include, but are not limited to the following: Does the doctor have a duty, legal or ethical, to tell the patient? Does the patient want to know? Was the patient aware of the possibility of an incidental finding? Is the doctor now responsible for that finding? What is the follow-up procedure? Should the finding be documented and, if so, how and where? How is the possibility of a false-positive result taken into account? Although the ultrasound literature has not yet addressed the ethics of incidental findings in models, magnetic resonance imaging (MRI) brain scan research literature has much to offer.
For the findings of brain MRI researchers, most doctors felt it was their ethical duty to inform the subject of an incidental finding. But some suggested that only the most certain of findings should be divulged because of the potential harm of a false-positive result. The same study shows that 97% of subjects surveyed said they did want to know of any incidental findings.4 As for the follow-up procedure, most again felt it was their responsibility to inform the model/research subjects and refer them to a doctor, with scans in hand, for further care. It should be noted that a significant minority felt that the follow-up should not be the entire responsibility of the subject.4
Documentation can be tricky because models are employees, not patients, and therefore, they do not and probably should not have a health record with their employer, but rather with their primary care physician. It is suggested then that any written documentation of the incidental finding be sent with the patient to be given to her physician. It is also suggested that EPs should compile a list of referrals and give the model appropriate referrals, encouraging follow-up. But the employer should also securely store a copy of the ultrasound scan for 5 years for possible future reference. Lastly, the patient has the right to know of the possibility of an incidental finding. However, the right to choose whether or not she wants to have that incidental finding disclosed to her can be complicated.
While there are still questions to be resolved concerning incidental ultrasound findings, it is recommended that models be made aware beforehand of the potential for an incidental finding and that such findings will be disclosed to them, regardless of the severity or the physician’s certainty.5This agreement should be both verbal and written to ensure that the model understands the possibility of a finding and that the documentation shows that understanding. An example of this written disclaimer form can be found in the Addendum (online only). Models also need to be reasonably made aware of the potential costs, both financially and emotionally, of incidental findings.
LIMITATIONS
While every effort was employed to find articles on the topic of incidental findings in ultrasound models, it’s possible that an article does exist but was not uncovered in our literature search.
CONCLUSION
During a training session for endovaginal ultrasonography, an incidental finding was discovered on the model, prompting follow-up care that resulted in surgery. Future training sessions will discover similar findings in models, and the physicians participating need to be prepared to deal with benign, malignant, and false-positive findings. The authors recommend the following approach to handling incidental findings in live ultrasound models: (1) inform the models and have them acknowledge before the session the possibility of an incidental finding, that they will be informed of any findings and of the possible costs that may be associated; (2) if an incidental finding occurs, inform the model; (3) compile a list of referrals and give the model appropriate referrals, encouraging follow-up; (4) securely store copies of the scans for 5 years for future reference; (5) give written documentation and copies of the scans to the model to be given to her physician.
There are undoubtedly other unforeseen implications of incidental findings that have not been discussed here. By establishing these guidelines for how incidental findings will be handled, currently known problems can be minimized.
Footnotes
Supervising Section Editor: Seric S. Cusick, MD
Submission history: Submitted August 3, 2010; Revision received October 21, 2010; Accepted February 7, 2011
Reprints available through open access at http://escholarship.org/uc/uciem_westjem
DOI: 10.5811/westjem.2011.2.2054
Address for Correspondence: J. Christian Fox, MD
University of California, Irvine, Department of Emergency Medicine 101, The City Dr, Rte 128-01, Orange, CA 92868
E-mail: jfox@uci.edu
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Grivell RM, Reid KM, Mellor A. Uterine arteriovenous malformations: a review of the current literature. Obstet Gynecol Surv. 2005;;60:761–767. [PubMed]
2. Mills P, Joseph AE, Adam EJ. Total abdominal and pelvic ultrasound: incidental findings and a comparison between outpatient and general practice referrals in 1000 cases. Br J Radiol.1989;;62:974–976. [PubMed]
3. Barnett SB. Live scanning at ultrasound scientific conferences and the need for prudent policy.Ultrasound in Med Biol. 2003;;29:1071–1076. [PubMed]
4. Illes J, Kirschen MP, Edwards E, et al. Ethics: incidental findings in brain imaging research.Science. 2006;;311:783–784. [PMC free article] [PubMed]
5. Johns Hopkins Medicine. Plans for detecting and managing incidental findings associated with research imaging procedures. Johns Hopkins Medicine Web site. 2010. Available at:http://www.hopkinsmedicine.org/institutional_review_board/guidelines_policies/guidelines/incidental_findings.html. Accessed July 22.


