| Author | Affiliation |
|---|---|
| Nima Majlesi, DO | Department of Emergency Medicine, Morristown Memorial Hospital, Morristown, NJ |
| Richard Shih, MD | Department of Emergency Medicine, Morristown Memorial Hospital, Morristown, NJ |
| Frederick W. Fiesseler, DO | Department of Emergency Medicine, Morristown Memorial Hospital, Morristown, NJ |
| Oliver Hung, MD | Department of Emergency Medicine, Morristown Memorial Hospital, Morristown, NJ |
| Renato Debellonio, MD | Department of Emergency Medicine, Morristown Memorial Hospital, Morristown, NJ |
ABSTRACT
Introduction:
Acute complications from cocaine abuse are commonly treated in the emergency department (ED); one of the most consequential is status epilepticus. The incidence of this complication is not clearly defined in the prior literature on cocaine-associated sequelae. We evaluated the incidence of status epilepticus in patients with seizures secondary to suspected cocaine use.
Methods:
We performed a retrospective multi-center study of patients with seizures resulting from cocaine use. We identified study subjects at 15 hospitals by record review and conducted a computer-assisted records search to identify patients with seizures for each institution over a four-year period. We selected subjects from this group on the basis of cocaine use and determined the occurrence of status epilepticus among them. Data were collected on each subject using a standardized data collection form.
Results:
We evaluated 43 patients in the ED for cocaine-associated seizures. Their age range was 17 to 54, with a mean age was 31 years; 53% were male. Of 43 patients, 42 experienced a single tonic-clonic seizure and one developed status epilepticus. All patients had either a history of cocaine use or positive urine drug screen for cocaine.
Conclusion:
Despite reported cases of status epilepticus with cocaine-induced seizures, the incidence of this complication was unclear based on prior literature. This study shows that most cocaine-associated seizures are self-limited.
INTRODUCTION
Cocaine intoxication has several emergency department (ED) presentations. Of those chief complaints, seizures have been considered one of the most consequential with a high morbidity and mortality.1 However, recent studies have shown that cocaine-associated seizures are commonly self-limited and may not require lengthy evaluation and work up in patients who have compete resolution of symptoms and a normal neurological examination.2–6 The incidence of cocaine-associated seizures is unclear. One study showed that among ED patients who had used cocaine, seizures were the chief complaint in approximately 8% of cases.5 Among pediatric patients with seizures, status epilepticus is one of the most serious complications with a short-term mortality of 8–20%.7,8
Although cocaine-associated status epilepticus has been reported in the literature, its incidence is unclear. The purpose of this study is to evaluate the incidence of status epilepticus in patients who present to the ED with cocaine-associated seizures.
METHODS
The study utilized a multi-center retrospective design. We identified patients with cocaine-associated seizures by retrospective record review over a four-year period at 15 hospitals, which included both urban and suburban teaching and non-teaching hospitals in New Jersey. In addition, the EDs included in this study used electronic medical records. We identified patients by ICD-9 code search for seizure or status epilepticus. After these records were identified, we performed a computerized search to identify cases associated with cocaine use. Cocaine use was defined as positive cocaine urine metabolites or a recent history of use within the previous 48 hours. Status epilepticus was defined as a seizure lasting longer than five minutes, or more than two discrete seizures with no intermittent period of normal consciousness (Figure 1).
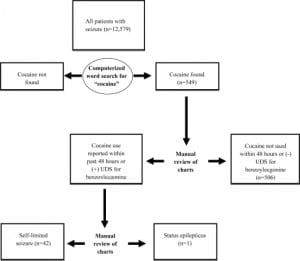
We collected data on each patient using a standardized closed-question data collection instrument. Information on patient demographics included age, gender, time from last illicit substance use, and urine toxicology results. Prior seizure history and past medical history was recorded. We also recorded results of head computed tomography (CT) scan, ethanol levels, electrolyte levels, and glucose. Drug co-ingestion was defined as an elevated alcohol level, a history of recent non-cocaine illicit drug use or positive drug testing of non-cocaine illicit drugs.
RESULTS
ED patient records from the 15 New Jersey hospitals totaled 1,590,629 over a four-year period between January 2002 and December 2006. All ED visits had digital medical records. Of these patient visits, we identified 12,579 seizure cases, and the word “cocaine” appeared in 549 patients’charts. Forty-three patients met the study inclusion criteria for cocaine-associated seizure. Of those with cocaine-associated seizures, 23 of 43 (53.5%) were male; 26 (60.5%) met criteria for co-ingestion; seven (16.3%) had prior history of a seizure disorder; and only one patient (2.3%) had status epilepticus. All study participants had generalized tonic-clonic seizures.
The sole patient with status epilepticus was a 41-year-old woman with a history of heavy ethanol and intranasal cocaine use over the previous day, who was found seizing by her mother. The seizures continued despite multiple doses of intravenous lorazepam and phenytoin, and she was paralyzed and intubated for approximately 24 hours. She was discharged without sequelae.
Of the other 42 cases, all had self-limited seizures that resolved while in the ED or prior to arrival. Three of these 42 patients had severe agitation after their self-limited episode of seizure activity. Two of these three were paralyzed and intubated but not actively seizing when rapid sequence intubation was performed. A CT scan of one showed a subarachnoid hemorrhage on CT scan, and one of the others was a body “stuffer” who ingested a large amount of cocaine while being arrested by law enforcement officials.
All 43 patients had normal sodium, calcium, magnesium, and glucose levels in the ED. Paramedics noted prehospital hypoglycemia in one patient with a blood glucose of 41 mg/dL. They treated the patient with glucagon, and he had a normal glucose level in the ED with complete resolution of seizure activity although no dextrose administration was documented. This patient also tested positive for phencyclidine on a urine drug analysis.
Twenty-six patients had documentation of past co-ingestion with other drugs of abuse or ethanol by history or urine drug screen. Ethanol was the most common co-ingestant, with 12 of 26 cases having detectable ethanol levels. Additionally, there were nine cases of opioids, eight of marijuana and one case of phencyclidine co-ingestion.
In 10 of 43 cases we documented routes of cocaine use, including nasal insufflation, ingestion, inhalation, and intravenous injection. Intranasal insufflation was the most commonly documented. Most patients (27 of 43) were discharged from the ED, although length of stay is unknown. Nine of these admissions had some neurologic or psychosocial sequelae ranging from agitation, psychosis, and suicidal ideation to lethargy and post-ictal states.
DISCUSSION
Status epilepticus secondary to cocaine use has been described in the medical literature. These cases are rare, predominantly documented in case reports, and associated with massive cocaine ingestions.9,10 Little information is available on the frequency of cocaine-associated seizures or status epilepticus. Further, the reported literature comes from EDs in urban settings.2–6 Our study attempted to identify patients with cocaine-associated seizures and to assess the incidence of status epilepticus using a 15-ED network in New Jersey. Yearly census for these 15 hospitals ranged from 24,000 to 83,000 patients with a mean of about 50,000. This ED network included both suburban and urban settings, covers approximately 15% of all EDs in New Jersey and may represent a better population cross segment than previously reportedly studies.2–6
The catchment area totaled over 1.5 million ED patient visits during the four-year study period. Despite the large number of patient visits, our study identified only 43 cases of cocaine-associated seizures, which included one case of status epilepticus. This rate is lower than in previous studies which were predominantly performed in urban EDs. Our study included mostly suburban EDs.2–6Although the retrospective study design may have failed to identify all patients with cocaine-associated seizures, the frequency of this event is likely to be low and the occurrence of status epilepticus even more uncommon. It is not clear why the incidence of cocaine-associated seizures is rare; however, it may be related to how they are caused. Experimentally, status epilepticus can be induced by single massive exposures to cocaine. Several reported human cases corroborate this mechanism.11 However, most cases of cocaine-associated seizures are likely due to the pharmacological phenomenon known as kindling, where repetitive administration of sub-convulsive doses of cocaine lead to seizure. This may be the reason for the low rate of cocaine-associated status epilepticus.12–14
The one patient we identified in our study was critically ill and difficult to manage medically, requiring multiple doses of benzodiazepines, paralysis and intubation. The role of ethanol co-ingestion is unclear but may have been a contributing factor. It is well known that cocaine and ethanol co-ingestion can lead to the formation of cocaethylene, a metabolite considered to be more cardio- and neurotoxic than those of cocaine alone. Similar to several previous reports, the majority of patients with cocaine-associated seizures cases did not involve status epilepticus.2–6 In addition, the majority of cases were managed without hospital admission.
LIMITATIONS
Our study was retrospective, lacked electroencephalogram testing and had limited follow up. And while cocaine is one xenobiotic often implicated in seizures, it is unclear if other potential ingestions may have been implicated. We did not perform confirmatory testing of other potential xenobiotics. In addition, we were unable to accurately assess the temporal relationship of cocaine use to seizure onset. Although some authors stress that cocaine-associated seizures occur early after use, especially within 90 minutes when peak concentrations of cocaine occur, the literature documents many cases occurring hours after drug use. Further, many of the study subjects had seizures witnessed by pre-hospital personnel upon arrival at the scene, although it is unclear how many. Thus, the diagnosis of seizure was often made by pre-hospital historical information. Further, we were unable to assess the duration of these pre-hospital seizures.
CONCLUSION
Status epilepticus is a rare event in patients presenting to the ED with cocaine-associated seizures. Most patients with this ED presentation were evaluated, treated and discharged.
Footnotes
The authors would like to acknowledge Timothy Grau, MD for the work that he contributed to this project. Additionally, this study was supported by a grant from the Emergency Medicine Associates Research Foundation
Supervising Section Editor: Jeffrey R Suchard, MD
Submission history: Submitted July 24, 2009; Revision Received November 16, 2009; Accepted November 29, 2009
Full text available through open access at http://escholarship.org/uc/uciem_westjem
Address for Correspondence: Richard D. Shih, MD, Morristown Memorial Hospital, 100 Madison Ave, Box #8, Depart. of EM, Morristown, NJ 07962
Email: shih100@yahoo.com
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Spivey WH, Euerle B. Neurologic complications of cocaine abuse. Ann Emerg Med. 1990;19:1422–8. [PubMed]
2. Holland RW, 3rd, Marx JA, Earnest MP, et al. Grand mal seizures temporally related to cocaine use: clinical and diagnostic features. Ann Emerg Med. 1992;21:772–6. [PubMed]
3. Pascual-Leone A, Dhuna A, Altafullah I, et al. Cocaine-induced seizures. Neurology. 1990;40:404–7. [PubMed]
4. Koppel BS, Samkoff L, Daras M. Relation of cocaine use to seizures and epilepsy. Epilepsia.1996;37:875–8. [PubMed]
5. Derlet RW, Albertson TE. Emergency department presentation of cocaine intoxication. Ann Emerg Med. 1989;18:182–6. [PubMed]
6. Alldredge BK, Lowenstein DH, Simon RP. Seizures associated with recreational drug abuse.Neurology. 1989;39:1037–9. [PubMed]
7. Lowenstein DH, Alldredge BK. Status epilepticus. N Engl J Med. 1998;338:970–6. [PubMed]
8. Chin R, Neville B, Peckham C, et al. Incidence, cause, and short-term outcome of convulsive status epilepticus in childhood: Prospective population-based study. Lancet. 2006;368:222–229.[PubMed]
9. Dhuna A, Pascual-Leone A, Langendorf F, et al. Epileptogenic properties of cocaine in humans.Neurotoxicology. 1991;12:621–6. [PubMed]
10. Conway EE, Jr, Mezey AP, Powers K. Status epilepticus following the oral ingestion of cocaine in an infant. Pediatr Emerg Care. 1990;6:189–90. [PubMed]
11. Winbery S, Blaho K, Logan B, et al. Multiple cocaine-induced seizures and corresponding cocaine and metabolite concentrations. Am J Emerg Med. 1998;16:529–33. [PubMed]
12. Miller KA, Witkin JM, Ungard JT, et al. Pharmacological and behavioral characterization of cocaine-kindled seizures in mice. Psychopharm. 2000;148:74–82.
13. Kaminski RM, Gasior M, Carter RB, et al. Protective efficacy of neuroactive steroids against cocaine kindled-seizures in mice. Eur J Pharm. 2003;474:217–22.
14. Kaminski RM, Witkin JM, Shippenberg TS. Pharmacological and genetic manipulation of kappa opioid receptors: Effects on cocaine-and pentylenetetrazol-induced convulsions and seizure kindling. Neuropharm. 2007;52:895–903.


