| Author | Affiliation |
|---|---|
| Laleh Gharahbaghian, MD | Department of Emergency Medicine, University of California, Irvine School of Medicine |
| Douglas P. Brosnan, MD, JD | Department of Emergency Medicine, University of California, Irvine School of Medicine |
| J. Christian Fox, MD, RDMS | Department of Emergency Medicine, University of California, Irvine School of Medicine |
| Samuel J. Stratton, MD, MPH | Department of Emergency Medicine, University of California, Irvine School of Medicine |
| Mark I. Langdorf, MD, MHPE | Department of Emergency Medicine, University of California, Irvine School of Medicine |
ABSTRACT
This case report describes an unusual presentation of an emergency department (ED) patient with nausea, vomiting, and epigastric pain, who was initially suspected of having viral hepatitis. The patient returned to the ED seven days later with persistent tachycardia and was diagnosed with new onset thyrotoxicosis.
INTRODUCTION
Thyrotoxicosis is an uncommon diagnosis in the emergency department (ED). Vomiting and abdominal pain are uncommon symptoms of thyrotoxicosis, and when coupled with laboratory abnormalities such as transaminitis, the emergency physician (EP) can be misled to a diagnosis of hepatitis.1 With thyrotoxicosis, many cellular metabolic processes are affected by excess thyroid hormone production, resulting in tachycardia, heat intolerance, palpitations, sweating, fatigue and tremulousness.1 Since thyrotoxicosis can progress to the life-threatening thyroid storm if untreated, early diagnosis with thyroid function tests and early intervention with beta blockers and agents to reduce thyroid hormone production and release are necessary. We present the case of a patient with vomiting, dehydration, and abdominal pain, which nearly resulted in exploratory laparoscopy, and in whom thyroid function tests showed new onset thyrotoxicosis.
CASE REPORT
A 28-year-old female physician presented to the ED with nausea and vomiting for three days with epigastric pain and decreased urination. The vomiting was non-bloody and non-bilious, occurred four times per day, and the patient was unable to tolerate liquids. The epigastric pain was mild, constant, and sharp. The patient felt a rapid pulse. She denied fever, chills, diarrhea, melena, or prior similar episodes. The past medical history was unremarkable with no prior surgeries, medical problems, or medications. The family history was documented as noncontributory and the patient denied pregnancy and alcohol, tobacco or drug use.
The physical exam revealed an oral temperature of 36.7° C, heart rate at 131 beats/min, respirations at 18 breaths/min, blood pressure at 128/75 mm Hg, and room air oxygen saturation (O2 sat) at 100%. The patient weighed 52.3 kg with a height of 157.5cm. She was alert and oriented, nontoxic and in no distress. The eye exam was normal without exophthalmos. There was no lymphadenopathy, no goiter was noted, and a thyroid exam was not documented. The lungs were clear and the cardiac exam was normal other than tachycardia. The abdomen was soft with normal bowel sounds, with mild tenderness in the epigastrium but no rebound or guarding. The neurological exam revealed normal cranial nerve function, full strength and sensation, and a normal gait. Deep tendon reflexes were not documented.
A metabolic panel including electrolytes, glucose, BUN and creatinine were normal. Liver function tests showed albumin at 3.6 g/dL(normal 3.2–5.5), AST at 118 IU/L(normal 8–40), ALT at 197 IU/L(normal 0–60), alkaline phosphatase at 51 IU/L(normal 26–110), and total bilirubin at 1.5 mg/dL(normal 0–1.4). The lipase level and complete blood count with differential were normal. A voided urinalysis showed specific gravity of 1.024, pH at 7.30, ketones at 150 mg/dL, small bilirubin, urobilinogen at 8 mg/dL, 2 red blood cells/high power field (hpf), 3 white blood cells/hpf, 6 epithelial cells/hpf, moderate bacteria, and no hemoglobin, leukocyte esterase, nitrite, protein, glucose, or crystals. A urine pregnancy test was negative. Bedside ED ultrasound by the EP showed no evidence of cholecystitis.
The patient received two liters IV normal saline and two doses of antiemetics. She then tolerated an oral liquid challenge, felt better and requested to be sent home. The discharge diagnosis was possible gastritis versus viral hepatitis. Lansoprazole 15 mg once daily was prescribed, with primary care physician (PCP) follow up in 1–2 days. A repeat set of vital signs was not documented.
Seven days later, the patient returned. She had seen her PCP who referred her for an esophagogastroduodenoscopy (EGD). She had not eaten or drunk for 20 hours prior to the procedure. The EGD, done with fentanyl and midazolam sedation, found a small hiatal hernia with no other abnormalities. Because of persistent tachycardia in the 140s/minute with intractable vomiting, the patient was sent directly to the ED. The patient stated the vomiting had resolved since the last ED visit and only started again after the EGD. The epigastric pain persisted without change and the only additional complaint was tremulousness. She continued to deny fever, chills, diarrhea, and melena.
The physical exam showed an oral temperature of 36.3° C, heart rate at 143 beats/min, respirations at 20 breaths/min, blood pressure at 125/61 mm Hg and O2 sat at 100% on 2L/min oxygen per nasal cannula. The patient weighed 50 kg (a 2.3 kg weight loss in one week). The patient was alert, tremulous and appeared fatigued. The remainder of the exam was unchanged. She received supplemental oxygen, three liters IV normal saline, and cardiac monitoring.
A repeat complete metabolic panel showed normal glucose and electrolytes except for bicarbonate at 19 mEq/L(normal 25–34), magnesium at 1.8 mg/dL(normal 1.8 – 2.5), albumin at 3.2 g/dL, AST at 83 IU/L, ALT at 87 IU/L, alkaline phosphatase at 48 IU/L, and total bilirubin at 0.8 mg/dL. A serum lipase level and complete blood count with differential were within normal limits. The erythrocyte sedimentation rate and C-reactive protein were normal. A urinalysis was not repeated. An EKG showed a narrow-complex sinus tachycardia at a rate of 152 beats/min with no ischemic ST or T wave changes. In addition, she had thyroid function tests sent, which are performed once daily by our institution’s laboratory. The patient tolerated oral liquids after antiemetics. Despite hydration, the patient’s tachycardia persisted at 130 beats/min. She appeared fatigued but had resolution of all other presenting symptoms. The patient was admitted to the internal medicine service on a telemetry floor.
Overnight she received two more liters IV normal saline, followed by maintenance rate. A right upper quadrant ultrasound showed a markedly thickened gallbladder wall without gallstones, a small amount of pericholecystic fluid, and a normal common bile duct. Radiology recommended a CT scan of the abdomen and pelvis, which showed small bilateral pleural effusions, periportal edema, upper abdominal ascites, marked gallbladder wall thickening, and moderate amount of free fluid in pelvis.
The next day general surgery recommended an exploratory laparoscopy. This procedure was delayed because the patient ate breakfast. The first two thyroid function panels sent from the ED and on hospital day two were invalid due to hemolysis, but a third set showed a free thyroxine (T4) level greater than 6 ng/dL(normal 0.8–1.9) and an undetectable thyroid-stimulating hormone (normal 0.5–5 mIU/L). After atenolol and methimazole, the patient improved and was discharged on the third hospital day with diagnoses of new onset thyrotoxicosis, dehydration, liver hemangioma, and small hiatal hernia.
DISCUSSION
Nausea, vomiting and abdominal pain are common complaints in the ED. However, they are uncommon signs of thyrotoxicosis.1 Thyrotoxicosis is uncommon itself, with a lifetime prevalence of 0.5% and an annual incidence of 30 cases per 100,000 persons per year. It occurs in a male-to-female ratio of 1:5–10.2 In British studies, the incidence is reported as high as 100–200 cases per 100,000 population per year.3,4 Although uncommon, it is important to detect thyrotoxicosis early due to the risk of life-threatening thyroid storm, which carries a mortality of 20–50% if untreated.5
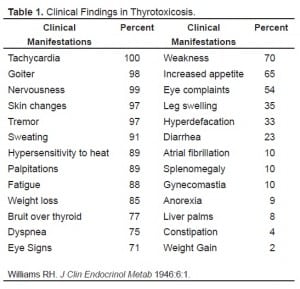
Patients with thyrotoxicosis typically present in a hypermetabolic state with sympathetic activation. The symptoms include anxiety or nervousness, psychosis, weight loss, excessive hunger, hyperdefecation, menstrual irregularities, pretibial edema, fever, heat intolerance, hyperhidrosis, goiter, ophthalmopathy (thyrotoxic stare/lid retraction), hyperreflexia, palpitations, tachycardia, hypertension, heart failure, and atrial fibrillation.6,7,8,9 A list of signs and symptoms in descending order of frequency is shown in Table 1. Our patient presented only with tachycardia, vomiting, and abdominal pain during her first visit, the latter two signs being uncommon in thyrotoxicosis. However, thyrotoxicosis has infrequently been associated with gastroparesis, resulting in abdominal pain and nausea. It was not until her return visit that she was noted to be tremulous.10
Like our patient, women in their third through sixth decade of life are at highest risk for thyrotoxicosis.6 Although our patient was not pregnant, excess exposure to thyroid hormone in utero is associated with high spontaneous abortion rates of up to 22%.11
Other rarely reported presentations of thyrotoxicosis include diabetic ketoacidosis in a previously well-controlled diabetic, near-fatal cardiac arrhythmias, and hypokalemic periodic paralysis.12,13 In addition, a previous case report discussed multi-organ dysfunction characterized by lactic acidosis and liver failure.14 Our patient did have an acidosis (bicarbonate 19 mEq/L) and although not in frank liver failure, did have elevated transaminases.
Since rapid thyroid function testing is unavailable in most EDs, the diagnosis of thyrotoxicosis is largely clinical. Persistent tachycardia, diffuse goiter, and hyperreflexia should sway the emergency physician to strongly consider thyrotoxicosis. Elevated levels of thyroxine (T4) and triiodothyronine (T3) with depressed thyroid-stimulating hormone (TSH) are diagnostic.6 Modest elevations in glucose (30–55% of patients), calcium (10% of patients), bilirubin, and transaminases (rare) may be seen, as well as a mild normochromic normocytic anemia.6 Our patient had evidence of dehydration on her urinalysis (specific gravity of 1.024) and chemistry panel (BUN/creatinine ratio greater than 20), but repeat vital signs during her initial visit were not documented to see if hydration had resolved her tachycardia. In addition, a family history was not documented which would have suggested the diagnosis in the ED, since it was later found that her mother had Graves’ disease.
The most common causes of thyrotoxicosis include diseases of primary hyperthyroidism with Graves’ disease as the most common cause (60–70%). This was our patient’s ultimate diagnosis by her endocrinologist. Other causes of primary hyperthyroidism include toxic multinodular goiter (15–30%) and iodine intake (rare). Thyroiditis (painful subacute, silent subacute, postpartum, radiation induced; 20%), central hyperthyroidism (pituitary adenoma; 3–5%), and nonthyroid diseases (ectopic thyroid tissue from stroma ovarii, metastatic thyroid cancer; rare) can also cause thyrotoxicosis. Drug ingestions and medication side effects precipitating thyrotoxicosis are rare.5
Drug-induced hyperthyroidism is linked to dietary supplements, especially metabolic boosters containing T3 or its derivatives, iodine-containing medications including indinovir and cough medications, and intentional or unintentional overingestion of levothyroxine. Iodine should also be avoided during imaging studies as this can worsen thyrotoxicosis. Our patient had a CT scan with iodine contrast without documented effects. However, the thyroid uptake scan could not be interpreted due to the use of iodine. Another series reported an epidemic of thyrotoxicosis, ultimately attributed to beef products contaminated with bovine thyroid hormone.16 Perhaps one of the most notorious medications associated with thyroid dysfunction is amiodarone. In a case study published in 2005, more than 27% of cardiac patients started on amiodarone had thyroid dysfunction; 7% were thyrotoxic.15
Management of a patient with thyrotoxicosis depends on the clinical presentation. Mild hyperthyroidism may be managed with judicious outpatient care, whereas moderate to severe thyrotoxicosis or thyroid storm requires emergent treatment. Initial therapy consists of stabilization: airway protection, oxygenation, intravenous fluids and cardiorespiratory monitoring.5 Fever should be managed with acetaminophen and cooling techniques. Dehydration should be addressed with appropriate fluid replacement. Ultimately, anti-thyroid medications and beta-blockers should be started while precipitating factors such as infection, trauma, cerebrovascular accident, myocardial infarction, general surgery, and medication reactions identified and managed accordingly.5
Following stabilization, the treatment goal is to inhibit hormone synthesis and release, prevent conversion of T4 to T3, and block hormone action in the periphery.6 Inhibition of synthesis is accomplished by thioamides [propylthiouracil (PTU), methimazole] which block thyroid peroxidase. PTU is preferred as it blocks peripheral conversion of T4 to T3.
Lithium and iodine both inhibit hormone release. However, lithium is difficult to titrate, and, therefore, iodine is preferred.5 In order to prevent iodine organification (Wolff-Chaikoff effect), thioamides should be given at least one hour before iodine therapy.5Peripheral conversion, accounting for 85% of circulating T3, is impeded by PTU, propranolol, and dexamethasone.6 The sympathomimetic effects of thyroid hormone are also managed with propranolol, which can reduce dysrhythmias, fever, tremors, palpitations, and anxiety.6 If clinical deterioration occurs in spite of appropriate therapy, thyroid hormone may be removed via exchange transfusion, plasmapheresis, or charcoal plasmaperfusion, the latter being cited in textbooks but rarely done in clinical practice.6Our patient was ultimately treated with methimazole and atenolol and showed clinical improvement prior to discharge.
Although our case is an unusual presentation of an uncommon disease, it is prudent to focus on the learning points from the initial ED visit. Our patient presented with vomiting, tachycardia, and a urine analysis consistent with her initial diagnosis of dehydration. Hypovolemia-induced tachycardia should resolve after adequate fluid resuscitation.17 During her second presentation, our patient was persistently tachycardic despite three liters of IV normal saline. Persistent tachycardia mandates a search for other causes including acute anemia, pulmonary embolism, arrhythmia, sepsis, toxic ingestions (sympathomimetic or anticholinergic agents), sedative/hypnotic withdrawal, hyperthyroidism, or other hyperadrenergic state. Further, a family history for autoimmune disorders suggests Graves’ disease. Our patient was also found to have free fluid in her pelvis and a thickened gall bladder wall, almost resulting in laparoscopy. Since there are no reports of gallbladder wall thickening associated with thyrotoxicosis, this finding is best explained by overhydration, which is a known cause of ascites and gallbladder wall thickening.18,19 Our patient received 5 liters IV normal saline within eight hours of her arrival and continued at a maintenance rate.
CONCLUSION
Thyrotoxicosis is an uncommon disease; however, its detection is important, as thyroid storm can develop if left untreated, carrying a mortality of 20–50%. Thyroid function testing is not available in most EDs. Therefore, diagnosis of thyrotoxicosis is largely clinical and relies on a thorough history and physical exam, including evaluation of family history. A repeat set of vital signs is necessary prior to discharging any patient, and a thorough evaluation for unexplained tachycardia is warranted. Thyrotoxicosis may present with abdominal pain and vomiting while other characteristic features are absent. The therapy for thyrotoxicosis consists of PTU, iodine and beta blockers. Moderate to severe thyrotoxicosis warrants admission to the hospital.
Footnotes
Submission history: Submitted May 10, 2007; Accepted June 29, 2007.
Full text available through open access at http://escholarship.org/uc/uciem_westjem
Address for correspondence: Mark Langdorf, M.D. MHPE, 101 The City Drive, Route 128, Orange, CA 92868
Email: milangdo@uci.edu
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Melmed S. Thyrotoxicosis. In: Dale DC, Federman DD, editors. ACP Medicine. 2006 Edition. New York: WebMD Professional Publishing; 2006. pp. 592–601.
2. American Association of Clinical Endocrinologists. Medical Guidelines for Clinical Practice for the Evaluation and Treatment of Hyperthyroidism and Hypothyroidism.Endocrine Practice. 2002;8:458–469.
3. Tunbridge WM, Evered DC, Hall R, et al. The spectrum of thyroid disease in a community: the Whickham survey. Clin Endocrinol (Oxf) 1977;7:481–493. [PubMed]
4. Vanderpump MP, Tunbridge WM, French JM, et al. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol (Oxf) 1995;43:55–68. [PubMed]
5. Tintinalli J, Kelen G, Stapczynski JS. Emergency Medicine: A Comprehensive Study Guide. 6th ed. McGraw-Hill: 2004.
6. Marx J, Hockberger R, Walls R, Wogan J. Rosen’s emergency medicine: concepts and clinical practice. 5th ed. Mosby; 2002. p. 2.
7. Parmar M. Thyrotoxic Atrial Fibrillation. Med Gen Med. 2005;7:74.
8. Brownlie B, Rae A, Walshe J, Wells J. Psychoses associated with Thyrotoxicosis. Eur J Endocrinol. 2000;142:438. [PubMed]
9. Williams RH. J Clin Endocrinol Metab. 1946;6:1. (text: Current Surgery)
10. Groskreutz JL, Kim CH, McConahey WM. Acute gastroparesis associated with thyrotoxicosis. Am J Gastroenterol. 1990;85:1206–1207. [PubMed]
11. Anselmo J, et al. Fetal loss associated with excess thyroid hormone exposure. JAMA.2004;292:691. [PubMed]
12. Loh K, et al. Thyrotoxic periodic paralysis complicated by near-fatal Ventricular Arrhythmia. Singapore Med J. 2005;46:88. [PubMed]
13. Bhattacharyya A, Woles P. Diabetic ketoacidosis precipitated by thyrotoxicosis.Postgrad Med J. 1999;75:291. [PMC free article] [PubMed]
14. Jiang Y, Hutchinson K, Bartelloni P, Manthous C. Thyroid storm presenting as multiple organ dysfunction syndrome. Chest. 2000;118:877. [PubMed]
15. Ross I, et al. Amiodarone-induced thyroid dysfunction. S African Med J. 2005;95:180.
16. Parmar M, Sturge C. Recurrent hamburger thyrotoxicosis. CMAJ. 2003;169:415.[PMC free article] [PubMed]
17. Merk Manual for Healthcare Professionals. Dehydration. 2005.http://www.merck.com/mmpe/sec19/ch276/ch276b.html.
18. Mayberry JC, et al. Mechanism of acute ascites formation after trauma resuscitation.Arch Surg. 2003;138:773–776. [PubMed]
19. Sanders RC. The significance of sonographic gallbladder wall thickening. J Clin Ultrasound. 1980;8:143–146. [PubMed]