| Author | Affiliation |
|---|---|
| Kristi A. Stanley, BA | Keck School of Medicine of the University of Southern California Medical Center |
| Mel Herbert, MD | Keck School of Medicine of the University of Southern California Medical Center |
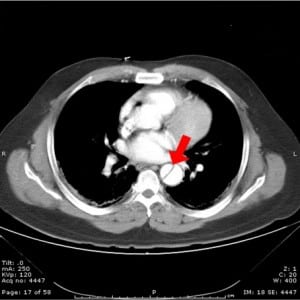
A 42-year-old patient presented with chest pain and shortness of breath. The pain was atypical for cardiac disease, started suddenly, occurred at rest and was non-radiating. Initial vital signs were normal and although the patient was in mild distress, the physical examination was normal. A Wells score was calculated and the patient was found to be in the moderate risk group for pulmonary embolism. A computed tomography pulmonary angiogram (CTPA) was performed (Figure 1).
The aortic flap seen (arrow) was unexpected but diagnostic. The patient was treated with beta-blockade and nitroprusside in the emergency room and went emergently to the operating room where the Stanford A aortic dissection was successfully repaired.
Aortic dissection is a relatively rare cause of chest pain but comes with high morbidity and mortality. Caused by stress on the aortic wall, the most common cause is longstanding systemic hypertension. Multiple indicators of dissection may be present during the physical exam, most characteristically the description of a “tearing” pain that radiates to the back. Other classic findings include a pulse deficit (25–30%), neurologic findings (5–17%), or a history of syncope (8–13%). While most patients are hypertensive, the presence of hypotension should raise the concern that the dissection may extend backwards into the pericardium.
Classic findings on chest x-ray include mediastinal widening, obliteration of the aortic knob, right-sided deviation of a nasogastric tube, depression of the left mainstream bronchus, or a small left-sided pleural effusion. In the stable patient, contrast CT scanning will show the extent of the dissection and pericardial and pleural effusions. However, CT scan does not precisely localize intimal tears, reliably demonstrate side branch involvement, or define aortic regurgitation. In the unstable patient, bedside transesophageal echocardiography (TEE) is used to find proximal aortic dissections and assesses for aortic insufficiency, pericardial effusions, and myocardial infarction. However, TEE requires an experienced operator and does not allow for evaluation of the entire descending aorta or side branch arteries.
Treatment of aortic dissection is dependent on the anatomy of the dissection, and two classification schemes are used. The Stanford classification labels the dissection as “A” if the ascending aorta is involved or “B” if the descending aorta is involved. The DeBakey classification system assigns labels of I for involvement of both the ascending and descending aorta, II for ascending aorta involvement, or III for descending aorta involvement. Treatment begins with controlling blood pressure with short acting, titratable medications. βblockade is used to decrease the stress the systolic pulse places on the aorta, and nitroprusside can be used to control hypertension. In the case of ascending aortic dissection, surgical replacement of the dissected segment is necessary. During this procedure, aortic insufficiency and coronary insufficiency can be corrected case of a descending aortic dissection, treatment utilizes intensive blood pressure control but may become surgical should uncontrollable hypertension, rupture, or involvement of a major aortic branch with subsequent end-organ ischemia occur. Another option for treating descending aortic dissections is stent-graft therapy, which provides an alternative that has been shown to be safer than surgery.
Footnotes
Submission history: Submitted July 17, 2007; Accepted August 10, 2007.
Full text available through open access at http://escholarship.org/uc/uciem_westjem
Address for correspondence: Lorien Hatch, Department of Emergency Medicine, LAC+USC Medical Center, Unit #1, Room 1011, 1200 N. State St., Los Angeles, CA 90033
Email: lhatch@usc.edu
Conflicts of Interest: By the WestJEM article submission agreement, all authors are required to disclose all affiliations, funding sources, and financial or management relationships that could be perceived as potential sources of bias. The authors disclosed none.
REFERENCES
1. Haro LH, Krajicek M, Lobl JK. Challenges, controversies, and advances in aortic catastrophes. Emerg Med Clin N Am. 2005;23:1159–1177.
2. Nienaber CA, Eagle KA. Aortic dissection: new frontiers in diagnosis and management: Part I: from etiology to diagnostic strategies. Circulation. 2004;109:E24. [PubMed]